Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
4.16 Increase in temperature increases the rate constant of a reaction. as we know increase in temperature increases the rate of reaction to satisfy the equation Rate = k [concentration]n where n can be any real number. k have to increase as concentration is almost not changing over small temperature change.
Increasing the temperature by 10°c almost doubles the rate constant. This can be represented quantitatively by the help of the Arrhenius equation-
K = Ae-Ea/RT, where k is rate constant, Ea is the activation energy, R is the universal gas constant, T is the absolute temperature.
New Question
11 months agoContributor-Level 10
5.6
Desorption is a process in which substance (reactant + product) is released from the surface which is the opposite process of sorption.
The role of desorption in the process of catalysis is to make the surface of the solid catalyst-free for fresh adsorption of reactants on the solid surface for further reactions to take place.
New Question
11 months agoContributor-Level 10
The complex is an anion with chromium as central atom, 2 water molecules and 2 oxolate ions with -2 negative charge Balance overall charge as 0, we get oxidation state of Cr as:
X + 2 (0) + 2 (-2) = -1 X = + 3.
Name of compound: potassium diaquadioxolatochromate (III) trihydrate. Electronic configuration of Cr: 3d3, t2g3
New Question
11 months agoContributor-Level 10
4.15 Let suppose reaction
A? B; having rate law, Rate = K [A]2
(i) If the concentration of A is doubled then the rate will affect by the square of concentration i.e. rate will become 22 = 4 times.
(ii) If the concentration of A is halved, then the rate will affect the square of the concentration i.e (1/2)2 = 1/4
(i) Rate becomes 4 times of initial Rate (ii) Rate becomes 1/4 times of initial Rate
New Question
11 months agoContributor-Level 10
(i) Overall charge balance:
X + 3 (-2) = -3 X = + 3
Oxidation state of Co is + 3.
As there are 3 oxolate ion and being bidentate, coordination no. Of complex is 6. So it is octahedral complex.
d orbital occupation: t2g6eg0 (oxolate ion is weak field ligand, does not cause pairing of electron as the energy required for pairing of electron is more than CFSE).
(ii) Overall charge balance:
X + balance4 (-1) = -2 X = + 2
Oxidation state of Co is + 2.
As there are 4 fluoride ion, coordination no. Of complex is 4 i.e. Tetrahedral complex.
d orbital occupation: eg4t2g3 (fluoride ion is weak field ligand, does not cause pairing of electron as th
New Question
11 months agoContributor-Level 10
4.14 Factors affecting rate of reaction-
1) Temperature (increasing temperature increases rate of reaction)
2) Concentration or pressure of reactants. (Increasing concentration or pressure increases rate of reaction)
3) Presence or absence of a (Adding catalyst mostly increases the rate of reaction)
4) The surface area of solid (If reaction is processing over solid reactant then increasing its surface area increases rate of reaction)
5) Nature of reactants
New Question
11 months agoContributor-Level 9
The qualifying requirement for admission to the BBA course at Dr. CV Raman University Bihar is that candidates must complete their Class 12 from a respective board of education from any stream. Moreover, for the admission process, the university releases the merit on the basis of candidates' achieved scores in Class 12 or also accepts CUET UG scores. To get admission, candidates can apply through the official website of the university.
New Question
11 months agoContributor-Level 10
5.5
Ester hydrolysis is represented as:
Ester + Water → Acid + Alcohol
In this reaction the acid produced which is a product also acts as a catalyst and makes the reaction faster.
Such substances that act as catalysts in the same reaction in which they are obtained as products are known as Autocatalysts.
So, ester hydrolysis is slow in the beginning and becomes faster after some time as more acid is produced on the product side.
New Question
11 months agoContributor-Level 10
4.13 Rate of given chemical reaction will be represented as
-d [pCH3OCH3] / dt
Hence units of rate is bar min-1
To find units of K, K = rate/ [pCH3OCH3]3/2
The unit of k = bar -1/2min-1.
Units of Rate- bar min-1. and Units of Rate constant K : bar -1/2min -1
New Question
11 months agoContributor-Level 10
Compounds containing carbonyl
Ligands only are known as homoleptic carbonyl. Such types of compounds are formed by most of the transition metals. These metal carbonyls always have simple, well-defined structures. In metal carbonyls the metal - carbonyl bond possess both s and p.character. M-C-bond is sigma bond. It is formed by the donation of lone pair of electrons of the carbonyl carbon into the vacant orbital of the metal. The M-C pi bond is formed by the donation of a pair of electron from a filled d orbital of a metal into the vacant antibonding? orbital of carbon monoxide. Such type of metal to ligand bonding creates a synergic ef
New Question
11 months agoContributor-Level 10
The colour of the particular complex compound depends on the crystal field splitting energy (CFSE). This CFSE depends on the nature of the ligand attached to the metal atom. In case of [Fe (CN)6]4– and [Fe (H2O)6]2+ the colour differs due to differences in CFSE .
CN- is a strong field ligand so will have high CFSE than H2O with a low value of CFSE. There is absorption of the energy from the visible region for the d-d transition and corresponding complimentary colour is observed. Thus there is the colour difference.
New Question
11 months agoContributor-Level 10
5.4
It is important to remove CO (Carbon Monoxide) in the synthesis of ammonia as CO affects the activity of Iron catalyst which is required in Haber's process.
Note: Haber's process is a very important industrial process which is used to produce ammonia.
New Question
11 months agoContributor-Level 10
In case of [Ni (H2O)6]2+ H2O is a weak field ligand, so it does not cause the pairing of the unpaired electron of Ni2+ ion. Thus there is possibility of the intra d-d transition from the d orbital of lower energy to that of higher energy. Thus the light is absorbed from the visible region and complimentary colour is observed. But in case of [Ni (CN)4]2– CN- is strong field ligand.
Therefore it will cause pairing of the unpaired electrons of Ni2+ ion. There are no unpaired electrons present, so there is no d-d transition and hence it is colourless.
New Question
11 months agoContributor-Level 10
5.3
As Adsorption is directly proportional to the available surface area and powdered form of a substance have a greater surface area than the crystalline form of the substance.
So, greater the surface area of the adsorbent more is the adsorption.
Hence, powdered substances are more effective adsorbents than their crystalline forms
New Question
11 months agoContributor-Level 10
Overall charge balance in [Cr (NH3)6]3+ complex:
X + 6 (0) = + 3 X = + 3
Cr is in + 3 oxidation state.
Electronic configuration of Cr in + 2 state: 3d3 . Now ammonia is a weak field ligand so it not causes pairing of the unpaired electron and undergoes hybridisation to form 6 sp3d2 hybrid orbitals filled by the six ammonia ligands. It's geometry is octahedral with unpaired electrons and hence is paramagnetic complex.
In case of [Ni (CN)4]2– ion :
Overall charge balance in [Ni (CN)4]2–complex:
X + 4 (-1) = -2 X = + 2
Ni is in + 2 oxidation state.
Electronic configuration of Ni in + 2 state: 3d8. Now cyanide ion is a strong field lig
New Question
11 months agoBeginner-Level 4
With a VITEEE rank of 212125, direct admission to VIT Vellore's main campus for popular core branches (like CSE, ECE, Mechanical) is highly unlikely because VIT Vellore cut-offs for these courses are usually below 30,000–50,000 for general category.
Even with your rank, you can still attend counselling — but you will most likely get branches in the Amaravati or Bhopal campuses, or less in-demand branches in Vellore/Chennai. Examples: Civil Engineering, Production, Biotechnology (if seats remain).
VIT offers a management quota where you can choose your preferred branch/campus regardless of rank — but the tuition fee is much higher,
New Question
11 months agoContributor-Level 10
The difference between the energies of the two set of the d orbitals is called as crystal field splitting energy (CFSE). The degenerate d orbitals split into two levels i.e t2g and eg level due to the presence of the ligands. This splitting of the degenerate orbitals due to the ligand is called as crystal field splitting and the energy difference between the two levels is called as crystal field splitting energy.
After the splitting of the degenerate orbitals has taken place the filling of the electrons takes place. Now first 3 electrons goes into the lower energy three t2g orbitals. The fourth electron can be filled in two ways:
It can
New Question
11 months agoContributor-Level 10
5.2
As Physisorption is Exothermic in nature, which means when gas gets adsorbed on the solid surface, Heat is evolved. So, according to Le-Chatelieres when the temperature is an increased reverse process (Desorption) will be favoured. So, Physisorption decreases with the increase of temperature.
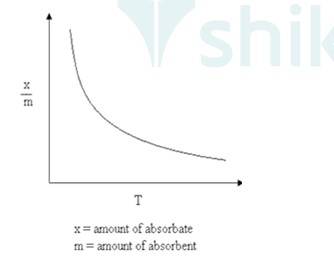
Where x/m: Volume of gas adsorbed
T: Temperature.
New Question
11 months agoContributor-Level 10
The strong ligands have higher splitting power of d orbitals of the central metal ion, whereas weak ligand has relatively lower splitting power of d orbitals of the central metal ion. The energy difference between t2g and eg sets of d orbitals is CFSE. The strength of the ligands depend on the magnitude of Δ . Strong ligands have larger value of CFSE and in case of weak ligands the CFSE values are smaller. The common ligands can be arranged in a series in the order of their decreasing field strength, as follows.
This series depends on the power of splitting the d orbitals and is called spectrochemical series, The order of fi
New Question
11 months agoContributor-Level 10
5.1
The two characteristics of Chemisorption are:
1. In Chemisorption which is highly specific in nature, the adsorb ate and adsorbent get attached by chemical bonds which are either covalent or ionic in
2. High activation energy is required and high temperature is also
3. Chemisorption increases with the increase in surface area which results in more number of active
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
