Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
Carbon due to its chemical property of catenation, tetravalency and the ability to form stable covalent bonds form hydrocarbon and halo-alkanes easily.
The process of removal of hydrogen and halogen from adjacent carbon atoms of haloalkens is called dehydrohalogenation. NaOEt ( Sodium ethoxide) used in Dehydrohalogenation. It is a strong base and helps remove a β-hydrogen from the haloalkane.
The chemical reaction for
Reaction:
R–CH2 –CHX–R′+NaOEt ethanol > R–CH=CHR’+NaX+EtOH
(i) 1-Bromo-1-methylcyclohexane:
Br
New Question
11 months agoContributor-Level 10
Cluster University select candidates based on performance in CUET-PG exam. The registrations for the entrance test are carried from Jan to Feb. CUET-PG exam is conducted from Mar to Apr. In the year 2025, the exam was conducted from Mar 13 to Apr 1. Additionally, the result was announced on May 6.
New Question
11 months agoContributor-Level 10
7.32
Helium mixed with oxygen under pressure is given to sea divers for respiration because pure oxygen can be toxic at a great concentration at the depth. Therefore oxygen can be mixed with helium to reduce oxygen concentration while eliminating nitrogen.
The main reason for adding helium to the breathing mix is to reduce the proportion of nitrogen and oxygen below those of air, to allow the gas mix to be breathed safely on deep dives.
A low proportion of nitrogen is required to reduce nitrogen narcosis and other physiological effects of gas at depth.
New Question
11 months agoContributor-Level 10
(i) Since I- ion is a better leaving group than Br- ion, therefore, CH3I reacts faster CH3Br in SN2 reaction with OH- ion.
Better the leaving group, faster is the SN2 reaction.
(ii) On steric grounds, 1? alkyl halides are more reactive than tert-alkyl halides in SN2 reactions. Therefore, CH3Cl will react at a faster rate than (CH3)3CCl in a SN2 reaction with OH- ion. (Bulkier the group, slower is the SN2 )
New Question
11 months agoContributor-Level 10
Hi, the eligibility criteria for best colleges for Game Design courses in India can vary depending on the specific programme and institution. However, here are some common requirements:
UG:
- Students must have completed Class 10+2 in any stream from a recognised board. Some institutions may also prefer candidates with a background in Science, Mathematics, or computers.
PG:
- Students must have a bachelor's degree in any discipline, although a degree in Computer Science, Animation, or a related field is often preferred.
- Some institutions conduct entrance exams to assess candidates' aptitude, creativity, and problem-solving
New Question
11 months agoContributor-Level 10
Ambident Nucleophiles are those nucleophilies which can attack through different sites. For example:-cyanide ions are a resonance hybrid of the following two structures:

It can attack through carbon to form cyanide and through N to form is O cyanide. Example: NO2, NO3 - etc.
New Question
11 months agoContributor-Level 10
If we compare the M.Tech programme of SVUCE, Rajalakshmi Engineering College (REC) and CIT Chennai, we will see that REC and CIT Chennai have the same tuition fee at INR 50,000 whereas, SVUCE M.Tech tuition fee is varied. The total tuition fee of the M.Tech programme is INR 37,800 to 86,000 depending on the type of course chosen.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
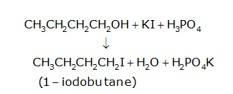
(a) 1-butanol is treated with KI in the presence of H3PO4 where the OH is being replaced by the iodine and gives H2O and KH2PO4 as the by-product with 1-iodobutane as the final
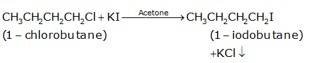
(b) The conversion of 1-chlorobutane to 1-iodobutane simply takes place by treating the reactant with KI in the presence of acetone. The iodine from KI normally replaces the chlorine from the reactant and gives 1-iodobutane as the final product with KCl as the by product.
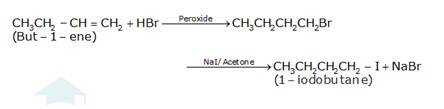
(c) The conversion of but-1-ene to 1-iodobutane takes place according to anti-markovnikoff (positive charged ion H+ goes to the carbon which has less number of hydrogens and negative part Br-
New Question
11 months agoContributor-Level 9
The total tuition fee for BSc courses at Dr. C.V. Raman University ranges from INR 30,000 to INR 2.4 lakh. On the other hand, the total tuition fee for BSc courses at KK University ranges from INR 40,000 to INR 2.8 lakh. Hence, it clearly can be seen that CVRU Bihar is more affordable than KK University for BSc courses. However, the fee structure can be only one component that candidates consider while comparing institutes, but several factors can be taken into account while choosing the best university/ college, such as placement packages, seats, ranking of that particular institute, affiliation, accreditation, etc.&nbs
New Question
11 months agoContributor-Level 10
Navrachana University offers BTech in four specialisations. One of these specialisations is Mechanical Engineering. Like all other specialisations, it is a four-year programme. The course curriculum is divided into eights semesters. It includes various theoretical and practical subjects. Some of these subjects are listed below:
- Experiential Engineering
- Physics for Mechanical Engineering
- Engineering Mechanics
- Strength of Material
- Thermodynamics
- Engineering Metallurgy
- Fluid Mechanics
- Kinematics and Dynamics of Machine
- Heat & Mass Transfer
- Energy Conversion
- Fluid Machinery
New Question
11 months agoContributor-Level 10
Double bond equivalent is used to find the level of unsaturations present in an organic molecule.
DBE = C +1-H/2+X/2-N/2
Where C= number of carbon atoms present H=number of hydrogen atoms present N=number of nitrogen atoms present X=number of halogen atoms present Double bond equivalent (DBE) for C4H9Br
= 4+1-9/2+1/2
=0
So none of the isomers has a ring or unsaturation, so the isomers are positions or chain isomers as shown below in the table:

New Question
11 months agoContributor-Level 10
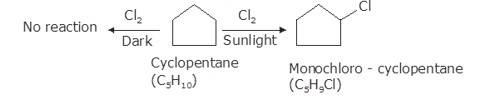
As per the molecular formula , one thing can be confirmed; the hydrogen is either cycloalkane or it is an alkene. However, alkenes readily react with chlorine in even dark because of the double bond. Therefore, it is possible that the hydrocarbon is cycloalkane.
Now, let us consider the reactivity. Cycloalkanes are the saturated hydrocarbons with no double bonds. These do not react with Chlore in dark because such a reaction needs UV light for initiating the process of free radical substitution. Haloalkane and Haloarenes NCERT solutions cover this as well as other questions in further detail.
In bright sunlight, the UV light star
New Question
11 months agoContributor-Level 10
Hi, the top entrance exams for admission to Game Designing colleges in India are NID Entrance Exam, NIFT Entrance Exam, CEED, Pearl Academy Entrance Exam, etc.
New Question
11 months agoContributor-Level 10
Dipole moment of a molecule depends on the electronegativity difference between atoms bonded covalently and geometry of the molecule (how far the atoms are from each other). Dipole moment is important to understand the polarity of a molecule.
The three dimensional structures of the three compounds along with the direction of dipole moment in each of their bonds are given below:-
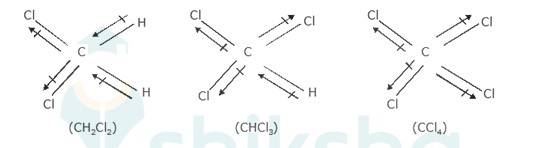
CCl4 being symmetrical has zero dipole moment. In CHCl3, the resultant of the two C-Cl dipole moments is opposed by the resultant of C-H and C-Cl bonds. Since the dipole Moment of latter resultant is expected to be smaller than the former, CHCl3 has a finite dipo
New Question
11 months agoContributor-Level 10
At TalentSprint Hyderabad, students are offered scholarships based on merit and need. However, it is important to note that the scholarships are limited in number. Thus, interested students must apply timely. The steps to apply are as follows:
1. Visit the official website of the institution.
2. Check the details of the preferred programme and find the scholarship details.
3. If eligible, connect with the RM assigned to you.
New Question
11 months agoContributor-Level 10
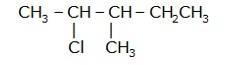
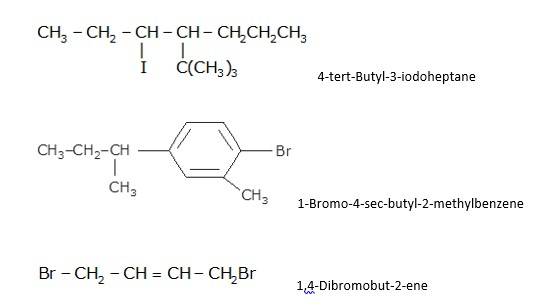
- Write the longest chain first e. in this case it is ‘pentane’ consisting of 5 carbons.
- Now, place the substituents at their respective place. For eg. Chlorine(Cl) at second carbon and methyl(CH3) at third carbon.
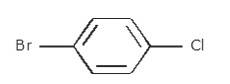
- Write the longest chain first i.e. in this case it is ‘benzene’ a cyclic structure consisting of 6 carbons
with 3 double bonds.
Now, place the substituent Bromine(Br) at the para position i.e. at the fourth carbon.
New Question
11 months agoContributor-Level 10
CH3CH (Cl)CH (Br)CH3: 2-Bromo-3-chlorobutane
- Write the name of side substituent according to the alphabetical order. Example: bromo is written before chloro as B comes before
- Always use a hyphen {-} between a number and a Since Br is attached to 2 position and Cl is attached to third position i.e. it is written as 2-Bromo and 3-chloro.
- Lastly, write the name of the longest hydrocarbon chain e. butane of 4 carbons.
CHF2CBrClF :1-bromo-1-chloro-1,2,2-trifluoroethane
- While writing the IUPAC name, write the name of side substituent according to the alphabetical Eg: bromo is written before chloro and fluoro as B comes before C and F.
- Always use a
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Yes, Talentsprint Hyderabad offers multiple scholarships for its students officially pursuing a certificate course. However, candidates must make sure that they fulfil the eligibility criteria set for the scholarship. As per the official website, the institute provides scholarships up to 30%. Moreover, women candidates are offered special scholarships. However, students must know the scholarships are limited in number and served on a first come, first served basis.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts