Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
Getting admission to TalentSprint Hyderabad Certificate courses depends on applicants' merit. The admission process designed for certificate courses starts with the application process. The application form can be obtained from the institute's official website.
Once the application window is closed, the institute reviews all the applications received. Those who meet the course-specific eligibility requirements are shortlisted for the selection stage. Further, the partner institutes make selections based on merit. Students who are offered admission might be required to report to the allocated institute for verification of documents and p
New Question
11 months agoContributor-Level 10

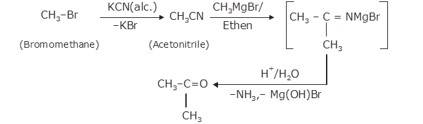
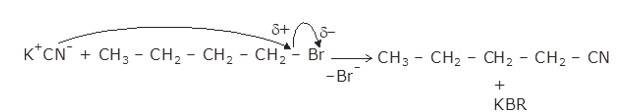
The given reaction is a nucleophillic reaction:

The given reaction is an SN2 reaction. In this reaction, CN acts as the stronger nucleophile and attacks the carbon atom to which Br is attached in nBuBr.
And, CN- ion is an ambident nucleophile and can attack through both C and N. In this case, it attacks through the C-atom.
The mechanism is shown below:
New Question
11 months agoContributor-Level 10
7.1
The general characteristics of Group 15 elements are:
Electronic configuration: All Group 15 elements have 5 electrons in their valence The general electronic configuration of these elements is ns2 np3
Oxidation state: Group 15 elements have 5 valence electrons and they require 3 more electrons to complete their However, the gaining of 3 electrons is difficult
Atomic size: Atomic size increases as we move down the group due to increase in the number of
Ionisation enthalpy: Ionisation enthalpy decreases as we move down the group because of increase in atomic
Electronegativity: Electronegativity decreases on moving down the group due to in
New Question
11 months agoContributor-Level 9
Yes, Class 12 marks are enough to get into the courses offered at Siva Sivani Degree College Hyderabad (SSDCH). The college provides four courses at undergraduate level. These programmes are BBA, BCA, BCom, and BSc. Since admissions are done on the basis of merit, candidates only need to provide the marks obtained by them in the last qualifying exam, i.e., Class 12. However, the college has a final say in deciding the final merit list.
New Question
11 months agoContributor-Level 10
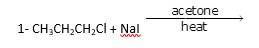
It is a simple substitution reaction with Cl being replaced by iodide ion.
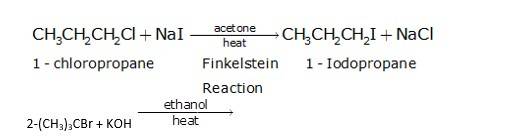
Since ethanol is a alcohol, so in presence of alcoholic KOH, alkyl chloride undergo elimination reaction, which results in the removal of proton and being substituted by a bromide ion.
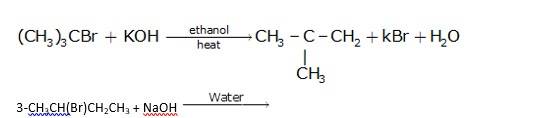
It is also a simple substitution reaction in which under the presence of aqueous NaOH, bromide ion is replaced by hydroxide ion as it is a better leaving group than hydroxide ion.
It is a simple substitution reaction in which a better leaving group leaves and is being substituted by an ion.
CH3CH2Br + KCN - CH3CH2CN + kBr
5-C6H5ONa + C2H5Cl
It is a simple substitution reaction in
New Question
11 months agoContributor-Level 10
7.34
It is difficult to study the chemistry of radon because it is a radioactive substance having a half-life (the time period to decompose the substance half to its initial concentration) of only 3.82 days.
Radon belongs to the 18 group elements with chemical formula as Rn. In general, the elements at the bottom o periodic table are radioactive, they are very dangerous to study as they emit harmful radiations.
Also, compounds of radon such as RnF2 have not been isolated, they are still in the phase of discovery. They have only been identified by radiotracer technique and no any further properties have been determined.
New Question
11 months agoContributor-Level 10
Navrachana University provides Merit-cum-Need Scholarships to BTech students who have scored a minimum of 70% in Class 12. Further, their family's combined income should not exceed INR 8 LPA. The amount of scholarship offered against various income slabs are presented in the table below:
| Scholarship | Family Income Slab |
|---|---|
| 100% Tuition Fee Waiver | Up to INR 2 lakh |
| 80% Tuition Fee Waiver | Between INR 2 Lacs to INR 3 lakh |
| 60% Tuition Fee Waiver | Between INR 3 Lacs to 4 lakh |
| 40% Tuition Fee Waiver | Between INR 4 Lacs to 6 lakh |
| 20% Tuition Fee Waiver | Between INR 6 Lacs to 8 lakh |
*Scholarship will be renewed every year provided the student maintains CGPA of 7 out of 10.
New Question
11 months ago
Contributor-Level 10
To get admission in Siva Sivani Degree College Hyderabad (SSDCH), candidates must first satisfy the course-specific eligibility criteria. As the college only offers undergraduate courses and admissions are purely merit-based. Interested students can apply for the desired course by visiting the college's website. For any admission-related queries, students can visit the official website of Siva Sivani Degree College Hyderabad (SSDCH) and fill out the enquiry form available.
New Question
11 months agoContributor-Level 10
(i) Freon 12:- the chlorofluorocarbon compounds of methane and ethane are collectively known as freons. They are extremely stable, unreactive, non-toxic and non-corrosive. Example of Freon 12 is CCl2F2. It is mainly used in refrigeration and eventually makes its way into the atmosphere where it diffuses unchanged into the stratosphere. In stratosphere, Freon is able to initiate the radical chain reactions that can upset the natural ozone balance.
- DDT: - it stands for p, p'-Dichlorodiphenyltrichloroethane (DDT). It is mainly used as an
- Iodoform: - it was earlier used as an antiseptic but the antiseptic properties are due the liberation o
New Question
11 months agoContributor-Level 10
Let us explain each question from Haloalkane and Haloarenes chapter one by one. Students who are currently in school and plan to take the CBSE board exam for class 12th soon, need to prepare all these questions. Let us get started.
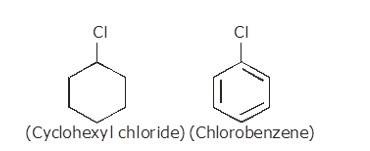
(i) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Sp2 hybrid carbon (s-character=33.33%) in chlorobenzene is more electronegative than a sp3-hybrid carbon (s-character=25%) in cyclohexylchloride, due to greater s-character. Thus, Carbon atom of chlorobenzene has less tendency to release electrons to Cl than carbon atom of cyclohexylchloride.
Chlorine atom in chlorobenzene is atta
New Question
11 months agoContributor-Level 10
Yes, BDes in Animation generally requires students to pass Class 12 from a recognised university with the required percentage for admission. Most of the popular government and private colleges in India demand at least 50% marks in Class 12 for accepting admission to the Bachelor of Design course in Animation.
Some top-ranking colleges may also ask for entrance exam scores as additional requirement for admission. The eligibility criteria varies for different colleges in India.
New Question
11 months agoContributor-Level 9
Shree Medha Degree College does not accept direct admissions for its courses. Although the selection criteria is merit-based, students must still satisfy the eligibility to get into the college. Candidates are advised to apply early as soon as the applications are made available to increase their chances at getting into the college. In case of any doubts, students can reach out to the admission help desk of the college.
New Question
11 months agoContributor-Level 10
To get a seat at CII Institute of Hospitality - ITC Kohenur for the Diploma courses, students need to complete a multi-step admission process which starts with application followed by selection and admission confirmation. Find below the steps to be eligible for admission at the institute:
- Eligibility: Pass Class 12 with minimum 50% aggregate
- Selection Criteria: Qualify CII Aptitude Test + Industry Panel Viva
New Question
11 months agoContributor-Level 10
The University of Wisconsin Milwaukee is one of the most-the finest universities for pursuing higher education in the United States of America. UW Milwaukee has a rich alumni network of more than 210,000 members all over the world. University of Wisconsin Milwaukee alumni network include innovators, leaders, mentors, entrepreneurs, famous personalities, and more. UW Wisconsin graduates work in top sectors such as Business Development, Information Technology, Finance, Sales, Engineering, Media and Communication, Marketing, and more. Furthermore, the University of Wisconsin notable alumni are mentioned below:
- Virginia Satir
- Steve Sisolak
- Ji
New Question
11 months agoContributor-Level 10
The University of Wisconsin Milwaukee is one of the oldest universities in the USA. The university has a rich alumni network all over the world. With more than 210,000 alumni members all over the world. UW Milwaukee alumni network include various famous personalities, innovators, industry leaders, game changers, and more. UW Milwaukee graduates work for top companies such as Amazon, Microsoft, Direct Supply, Kohler Co., Generac, and more. Some notable alumni at University of Wisconsin Milwaukee are Willem Dafoe, Golda Meir, Satya Nadella, Alberto Fujimori, John Edward Douglas, Trixie Mattel, Andy Hurley, Jack Kilby, and more. Furthermo
New Question
11 months agoContributor-Level 10
7.33
XeF6 + H2O XeO2F2 + HF
Balanced equation: XeF6 + 2H2O XeO2F2 +4HF. The steps for balancing the reaction are as follows:
1. The main element is First, check if the atoms of Xe on both sides are balanced. YES, they are.
2. Then take another element i.e. F. It is not balanced on the right side, there are 3 atoms of F missing. So, to balance it multiply HF by 4.
3. Now check the other secondary atoms which are oxygen and hydrogen. Oxygen is not balanced on the left side as there are 2 O atoms so multiply H2O by
4. Now check for the hydrogen They are balanced on both sides.
New Question
11 months agoContributor-Level 10
Candidates can check below benefits of pursuing a BSc Physiology:
Gain insights into how human body works at cellular and systemic levels.
Prepares you for advanced studies like MSc, Physiotherapy or even Medicine.
Offers many career options in diagnostics, rehabilitation and also wellness.
- Pursue roles in teaching, pharmaceuticals, sports science or even health consultancy.
Hope this helped your query! All the best!
New Question
11 months agoContributor-Level 10
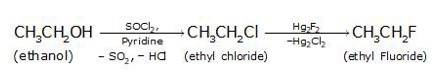
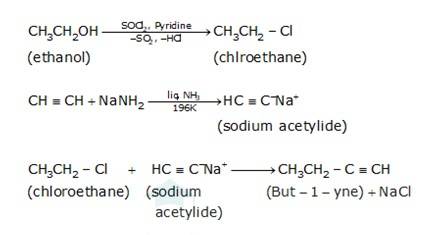
(i) Since the conversion of ethanol to but-1-yne involves the addition the two extra carbons that is why the conversion is carried out in 3 steps.
- In the first step ethanol is treated with thionyl chloride (SOCl2) in the presence of pyridine to give chloroethane
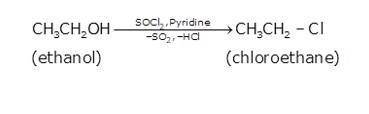
- In the second step the acetylene is treated with sodamide(NaNH2) in the presence of liquid ammonia to give sodium acetylide
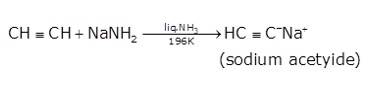
- The last step involves the reaction between chloroethane and sodium acetylide to give the final product as the But-1-yne and NaCl as the by-product.
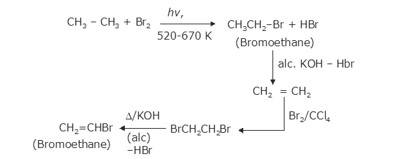
- The bromination of ethane (Br2 in the presence of light at 520-670K) gives bromoethane which on further treatme
New Question
11 months agoContributor-Level 10
The TalentSprint Hyderabad maintains a good network of recruiters. The list of eminent companies that participated in TalentSprint Hyderabad placements in recent years is presented below:
TalentSprint Hyderabad Top Recruiters | |
|---|---|
Genpact | EY |
Accenture | Cognizant |
Airtel | Microsoft |
TCS | Wipro |
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - ITC Kohenur Diploma eligibility requires students to pass Class 12 or equivalent with a minimum aggregate of 50% from a recognised board with English as a compulsory subject. In addition to fulfilling the eligibility, students must also clear the aptitude test and Industry-Panel Viva to get shortlisted. Interested students can head over to the official website of the college for more information.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts