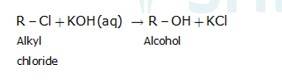Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
The MEXT Scholarship is the most prestigious and fully funded, covering tuition, living costs, and airfare.
New Question
11 months agoContributor-Level 9
Siva Sivani Degree College course-fee ranges between INR 75,000 and INR 2 Lakh. The fee varies depending on the course selected by the student. Check out the following table to know about Siva Sivani Degree College fees for the offered programmes:
COURSE | COURSE FEE |
|---|---|
| BBA | INR 2lakh |
| BCA | INR 1.1 lakh |
| BCom | INR 85,000 |
| BSc | INR 75,000 |
Note: This information is taken from the official sources and is subjective.
New Question
11 months agoContributor-Level 10
7.42
To draw the resonating structure of NO2 No. of valence electrons for N = 5
No. of valence electrons for O = 2 × 6 = 12
Total no. of valence electrons = 5 + 12 = 17
The N and O atoms are arranged in such a way that the less electronegative atom N is placed as the central atom.
Electron pairs are placed between bonds and distributed around the atoms to form octet as shown. Both the O atoms have their complete octet but N has only 5 electrons.

A pair of electrons is transferred from O to the bond between O and N. Both the O atoms have their complete octets and N has 7 electrons.

The resonating structures of NO2 can be shown as:

New Question
11 months agoContributor-Level 10
BSc Agriculture programme at Nagaland University – Medziphema Campus is a four-year course divided into eight semesters. It includes 176 credit hours in total, 136 for regular coursework and 40 under the Student READY Programme. The READY segment further breaks into 20 credit hours for ELP and 20 for AIA & RAWEP (10+10 each).
New Question
11 months agoNew Question
11 months agoContributor-Level 9
No, there is no entrance exam required for admission to Siva Sivani Degree College Hyderabad (SSDCH). Admission to all the courses is merit-based at this college. Candidates are required to apply to the official website of the institute and fill out the admission form. After the process is completed, the final seat allocation is based on the last qualifying exam score.
New Question
11 months agoContributor-Level 10
7.41
Copper metal on reaction with HNO3 gets oxidized and give different by-products depending on the temperature, concentration of the acid and the copper metal undergoing oxidation.
The reaction of copper with concentrated and dilute HNO3 is as shown below:
3Cu + 8HNO3 (did.) 3Cu (NO3)2 + 2NO +4H2O
Cu + 4HNO3 (conc.) Cu (NO3)2 + 2NO2 +2H2O
New Question
11 months agoContributor-Level 10
No, TalentSprint Hyderabad does not provide any refund in case a candidate wishes to withdraw his/her admission from any programme either before the commencement of the session or subsequent to the commencement of the sessions. As per the official website, the institution reserved the absolute and sole right to either accept or reject any request for refund of fees paid already for the current/future batch.
Moreover, if the institution terminates any participant from attending the further sessions, then the institution reserves the right to withhold from the fees of the participant an amount proportionate to the service already rendered
New Question
11 months agoContributor-Level 10
This is a classic question from the chapter Haloalkanes and Haloarenes. Let us solve each one as follows:
(1) When n - butyl chloride is treated with alcoholic KOH, the formation of but - l - ene takes place. This reaction is a dehydrohalogenation reaction.
When N-butyl chloride or 1-chlorobutane is treated with alcoholic potassium hydroxide (KOH), elimination reaction takes place. This leads to alkene formation. 1-butene is the major product here.
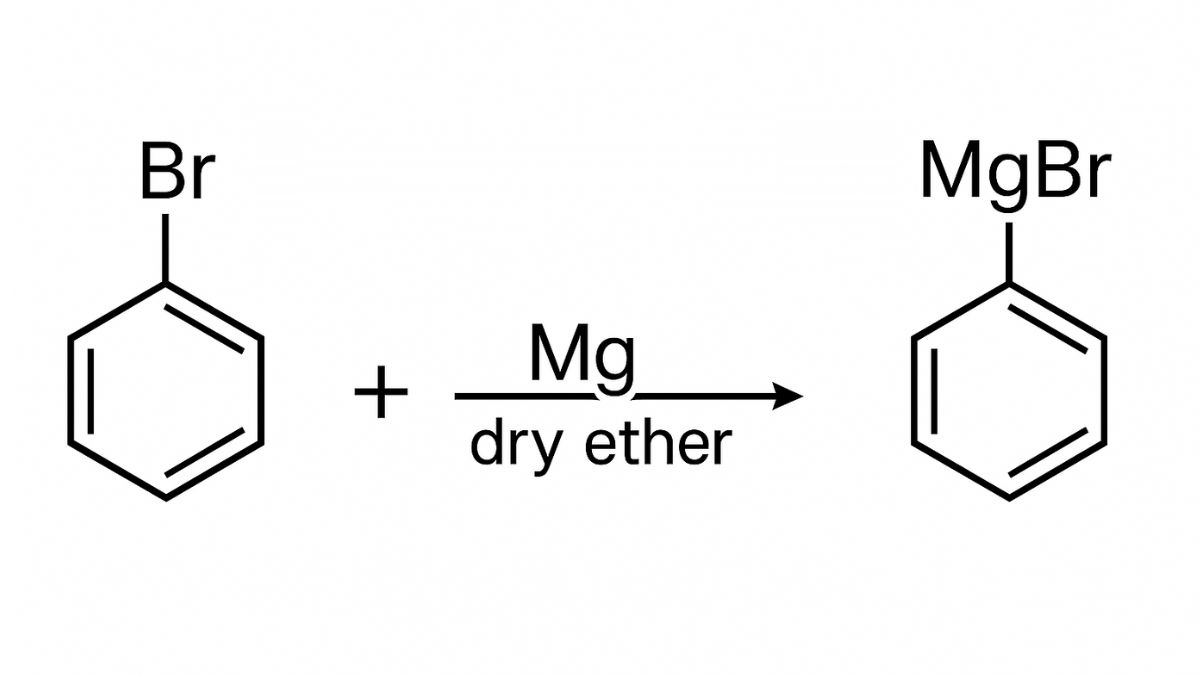
(ii) When Bromobenzene is treated with Mg in the presence of dry ether, it undergoes a reaction to produce phenylmagnesium bromide which is the Grignard reagent. This reagent is very re
New Question
11 months agoContributor-Level 6
To apply for CII Institute of Hospitality - ITC Grand Central, Mumbai, candidates must appear for the CII Aptitude Test, followed by an Industry-panel Viva. The application process is conducted in an online mode. Interested candidates can visit the official website to apply for the desired course. The minimum eligibility requirement is to pass Class 12 from a recognised board in India with a minimum of 50% in English.
New Question
11 months agoContributor-Level 10
7.40
Ammonia is manufactured industrially by Haber's process. Nitrogen from gas is combined with hydrogen derived from natural gas (methane) in the ratio 1:3 giving rise to ammonia. The reaction is reversible and exothermic.

The optimum conditions for the manufacturing of ammonia are pressure 200*105 Pa, the temperature of 4700 K and iron oxide catalyst with a small amount of Al2O3 and K2O.
New Question
11 months agoNew Question
11 months agoContributor-Level 9
The first step in the application process of Siva Sivani Degree College Hyderabad (SSDCH) is to visit the official website of college. Candidates are required to register by entering basic details or using a valid email ID and phone number to apply for admission.
New Question
11 months agoContributor-Level 10
Yes, there are about 10+ Law colleges in Chennai offer LLB course. Some of them are mentioned below along with their tuition fees:
| Top Colleges | Tuition Fee |
|---|---|
| SIMTS | INR 15 lakh |
| Tamil Nadu Dr Ambedkar Law University | INR 1.26 lakh |
| Dr. Ambedkar Government Law College, Chennai | INR 3,000 |
| SRM School of Law | INR 6 lakh |
Disclaimer: This information is sourced from official website and may vary.
New Question
11 months agoContributor-Level 10
Candidates wanting to enrol in MBA course of Cluster University need to apply for CUET-PG. Hence, the aspirants must complete the application process of the entrance exam. Table given below presents category-wise CUET-PG application fees:
Category | Application Fee (For upto two test papers) |
|---|---|
General | INR 1,400 |
Gen-EWS/ OBC-NCL | INR 1,200 |
SC / ST / Third Gender | INR 1,100 |
PwD | INR 1,000 |
New Question
11 months agoContributor-Level 10
There are two primary alkyl halides having the formulaC4H9Br, They are n - butyl bromide and isobutyl bromide.
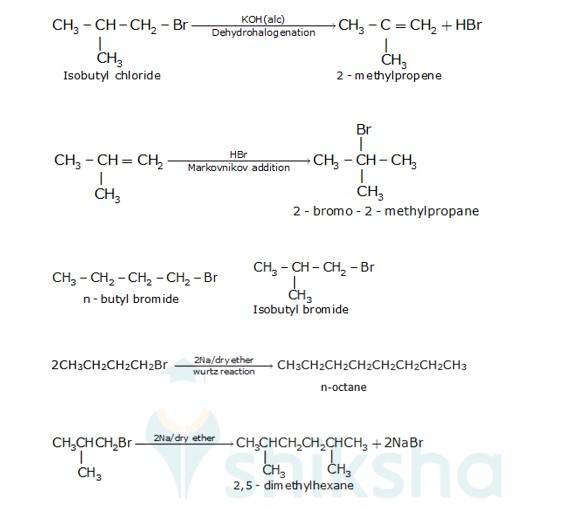
Therefore, compound (a) is either n-butyl bromide or isobutyl bromide.
Now, compound (a) reacts with Na metal to give compound (b) of molecular formula, C18H18 which is different from the compound formed when n-butyl bromide reacts with Na metal.
Hence, compound (a) must be isobutyl bromide. Thus, compound (d) is 2, 5-dimethylhexane.
It is given that compound (a) reacts with alcoholic KOH to give compound (b). Hence, compound (b) is 2- methylpropene.
Also, compound (b) reacts with HBr to give compound (c) which is an isomer of (a)
New Question
11 months agoContributor-Level 10
7.39
Nitrogen is prepared in the laboratory by heating aqueous ammonium chloride (NH4Cl) with sodium nitrite (NaNO2) to form ammonium nitrite (NH4NO2), which is unstable. Ammonium nitrite breaks down to form nitrogen and water.
NH4Cl (aq) + NaNO2 (aq) - NH4NO2 + NaCl (aq)
NH4NO2 - N2 (g)+ H2O (l)
Small amounts of NO and HNO3 are also produced which can be removed by passing nitrogen gas through aqueous sulphuric acid containing potassium dichromate.
New Question
11 months agoContributor-Level 6
No, you cannot join CII Institute of Hospitality, Mumbai directly. Admission is based on the in-house entrance test, CII Aptitude Test, followed by an Industry-panel Viva. The candidates must meet the eligibility criteria set by the institution. Interested candidates can apply online on the official website for the desired course.
New Question
11 months agoContributor-Level 10
For admissions to BSc courses at Dr. C.V. Raman University, candidates are required to complete their Class 12 in PCB/PCM subjects. The subject differentiation differs according to the chosen specialisations. Moreover, for some specialisations, the university accepts candidates only from PCB streams or PCM/PCB streams. In addition, for admission to the BSc (Hons) in Mathematics, candidates from only the PCM stream can apply.
New Question
11 months agoContributor-Level 10
An aqueous solution, KOH almost completely ionizes to give OH-ions. OH- ion is a strong nucleophile (see the imp. note below), which leads the alkyl chloride to undergo a nucleophilic substitution reaction to form alcohol.
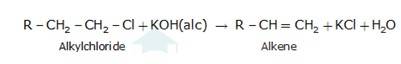
On the other hand, an alcoholic solution of KOH contains alkoxide (RO-) ion, it is a strong base. Thus, it can abstract a hydrogen ion from the beta-carbon of the alkyl chloride and form an alkene by eliminating a molecule of HCl.OH- ion is a much weaker base than RO- ion. Also, OH- ion is highly solvated (because more energy is released on solvation)in an aqueous solution and as a result, the basic character of OH-io
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts