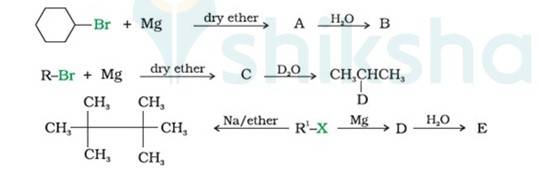Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
(i) 2-chloro-3-methylbutane, (2? alkyl halide)
Explanation: The Cl atom is attached with two alkyl groups therefore 2? alkyl halide.
(ii) 3-chloro-4-methylhexane (2? alkyl halide)
Explanation: The Cl atom is attached with two alkyl groups therefore 2? alkyl halide.
(iii) 1-iodo-2,2-dimethylbutane (1? alkyl halide)
Explanation: The atom I is attached with one alkyl group therefore 1? alkyl halide.
(iv) 1-bromo-3,3-dimethyl-1-phenylbutane (2? benzylic halide)
Explanation: The atom Br is attached with two alkyl groups with the benzene group therefore 2? benzylic halide.
(v) 2-bromo-3-me
New Question
11 months agoContributor-Level 10
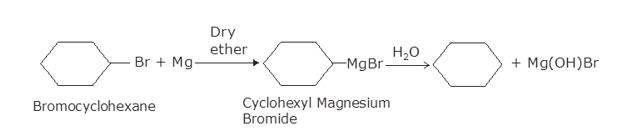
(i) Bromocyclohexane is reacting with the metal Mg to give organo-metallic compound/Grignard reagent
i.e. Cyclohexyl Magnesium Bromide (A).
Grignard reagents are highly reactive with hydrocarbons to give alkanes. Cyclohexyl Magnesium Bromide reacts with water to cyclohexane (B) and Mg (OH)Br.
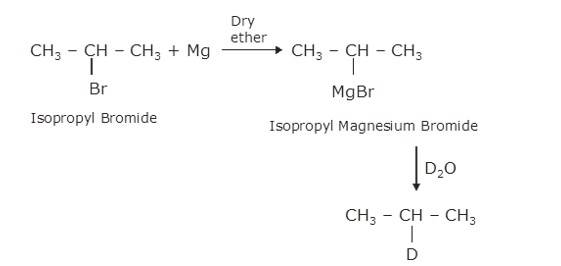
(ii) As the above reaction, haloalkane with metal Mg react to form the Grignard reagent, alkyl magnesium halide, RMgX, and further with hydrocarbon gives alkane.
∴ C is CH3CH (MgBr)CH3, a Grignard Reagent, R is CH3CH (Br)CH3 and D ≅ H
(iii) In the Wurtz Reaction, alkyl halide in the presence of sodium in dry ether gives the double number
New Question
11 months agoContributor-Level 10
Candidates seeking BTech admission at Navrachana University through the counselling conducted by the Admission Committee for Professional Courses (ACPC) are required to clear the cut-off to secure a seat. The cutoff is based on the applicants' scores/ ranks in GUJCET (Round 1) and JEE (Main) (Round 2). The cut-off varies across the years and the specialisations. The cut-off for the current year is not available as of now. In the year 2021, the GUJCET cut-off for various BTech specialisations ranged from 12,649 to 30,817.
New Question
11 months agoContributor-Level 10
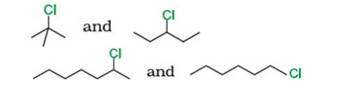

Reason: SN1 occurs in two steps. In the first step, carbocation forms. The rate of reaction in SN1 depends upon the stability of the carbocation, greater the stability faster the reaction. Tertiary (3°) carbocation is more stable than secondary (2°), which is further stable than primary (1°)
By using this
- Tert-Butyl Chloride (3°) reacts faster than 3-Chloropentane (2°)
- 2-Chloro heptane (2°) is more reactive than 1-Chloro hexane (1°)
New Question
11 months agoContributor-Level 10
Yes, of course. All popular colleges across India offering the BTech in CSE course provide excellent placement opportunities to graduates in Computer Science Engineering, extending healthy salary packages to students with the right amount of programming knowledge, skills, and internship experiences. You must work on sharpening your skill set to stand out in front of the recruiter, as the competition in the job market is high, and it is only a strong skill set that can make you get your dream job.
New Question
11 months agoContributor-Level 10
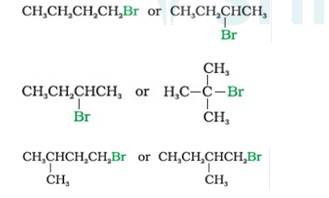
In SN2 Reaction, the product will be formed in a single step with no intermediates. Nucleophile attack will be easier for simple halides than hindered haloalkanes. Therefore,

- Bromobutane (1°)reacts faster than 2-Bromobutane (2°)
- 2-Bromobutane (2°) reacts faster than 2-Bromo-2-methylpropane or tert-Butyl bromide (3°) 1-Bromo-3-methyl butane reacts faster than the 1-Bromo-2-methyl bu
- tane as the former is less hindered with respect to leaving halide than the later which is more hindered comparatively
New Question
11 months agoNew Question
11 months agoContributor-Level 6
Yes, GMAC offers GMAT exam in two formats -
- Traditional: Offered at a GMAT exam centre
- From Home: Offered online, can be taken from home. Referred to as GMAT Online
Applicants have the choice to take GMAT either traditionally or online. The catch is that the fee varies for both the formats of GMAT exam. GMAT at test centre costs USD 275 wheeas GMAT Online costs USD 300.
New Question
11 months agoContributor-Level 10
The order of increasing boiling point is
- Chloromethane (CH3Cl)< Bromomethane (CH3Br) < Dibromomethane (CH2Br2)< Bromoform (CHBr3)
- Isopropyl chloride (C3H7Cl)< 1-Chloropropane (C3H7Cl) < 1-Chlorobutane (C4H9Cl)
Generally, boiling point increases with the molecular weight of the compound and decreases with the branching of the chain
New Question
11 months agoContributor-Level 10
Yes, there are various government job opportunities after completing BSc courses from Dr. C.V. Raman University, Bihar. Government jobs remain in high demand due to various benefits, including job security, competitive pay, tax benefits, and more. Students with a bachelor's degree in Science can apply for many different jobs in the public sector.
Moreover, studying a course like BSc improves a candidate's technical knowledge and analytical skills, which are necessary to crack government exams. Students with Science degrees can take government exams to seek research-based job opportunities at national institutes and laboratories. Tabulat
New Question
11 months agoContributor-Level 10
For admission to the TalentSprint AI for Leaders certificate course, students must fulfil the eligibility requirements set by the partner institute. Check below to know the eligibility criteria to be met:
- Applicants who have completed a graduation or postgraduate degree in any discipline with a minimum of 50% marks are eligible to apply
- Applicants should be working professionals or self-employed
- Minimum 10 years of work experience with an impactful career path
New Question
11 months agoContributor-Level 10
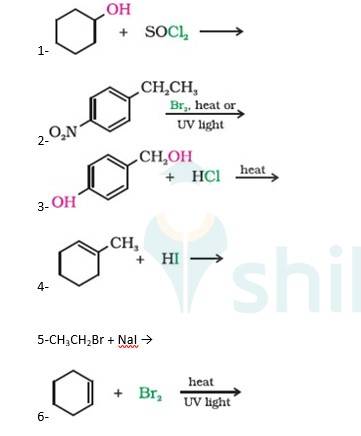
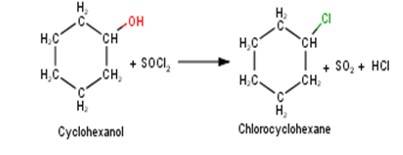
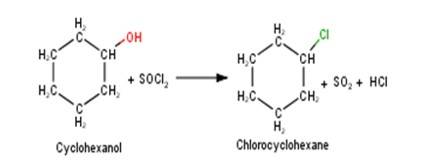
A 10.5 1. Cyclohexanol will react with thionyl chloride to form Chlorocyclohexane with the evolution of sulphur dioxide and hydrogen chloride gas as shown below :
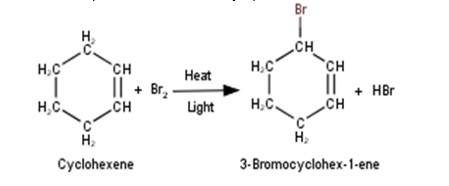
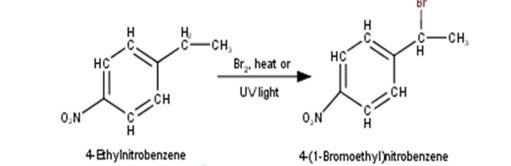
2. 4-Ethylnitrobenzene will undergo benzylic bromination in presence of heat or light. Now, as benzylic radicals are more stable therefore benzylic hydrogen is abstracted. Hence, the reaction yields 4-(1- Bromoethyl)nitrobenzene as a product
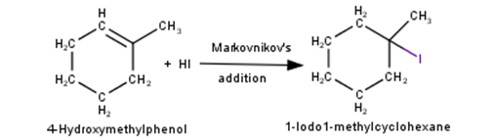
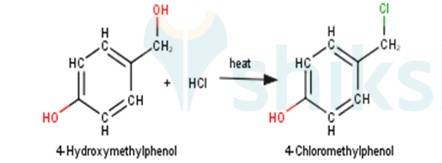
3. 4-Hydroxymethylphenol will react with hydrochloric acid under thermal conditions to yield 4- Chloromethylphenol a shown in the reaction below:
4. 1-Methylcyclohexene will react with hydrogen iodide via markovnikov’s addition mechani
New Question
11 months agoContributor-Level 10
Navrachana University BTech seats are offered to eligible candidates in two ways. Aspirants can either secure a seat through the ACPC (GUJCET) counselling based on GUJCET/ JEE (Main) scores or apply directly at the university.
Candidates willing to secure a seat for BTech at Navrachana University can follow the steps given below:
- Check your eligibility against the eligibility criteria laid down the university for BTech admission.
- Appear for GUJCET or JEE (Main).
- Register for admission through the official ACPC portal and select Navrachana University during choice filling (for 50% of the seats). For the remaining 50% of the seats,
New Question
11 months agoNew Question
11 months agoContributor-Level 10
If we compare SVUCE, Rajalakshmi Engineering College and CIT Chennai, we can see that SVUCE offers more M.Tech programmes than the other two colleges. Rajalakshmi Engineering College offers 11 M.Tech courses whereas Rajalakshmi Engineering College only offers eight and CIT Chennai only offers three courses. SVUCE also has a more varied tuition fee, with total tuition fee of the programme at INR 37,000 - 86,000 depending upon the course chosen.
New Question
11 months agoContributor-Level 10
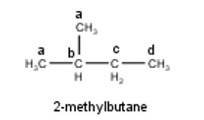
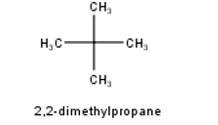
To have a single monochloride, there should be only one type of H-atom in the isomer of the alkane of the molecular formula C5H12. This is because of the fact that replacement of any H-atom leads to the formation of the same product. Therefore the isomer is 2,2-dimethylpropane.
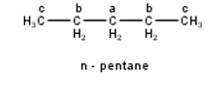
To have three isomeric monochlorides, the isomer of the alkane of the molecular formula C5H12 should contain three different types of H-atoms. Therefore, the isomer is n-pentane. It can be observed that there are three types of H atoms labelled as a, b and c in n-pentane as shown below : -
To have four isomeric monochlorides, the isomer of the alkane of the molecu
New Question
11 months agoContributor-Level 10
10.3 There are four different dihalogen derivatives of propane. The structures of these derivatives are as shown below: -
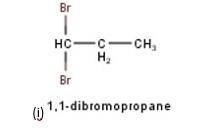

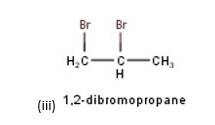
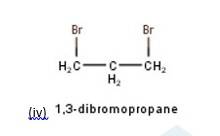
New Question
11 months agoContributor-Level 10
Once the shortlisting process is completed, selected students may be required to report to the allocated institute and present original and self-attested photocopies of documents for verification purposes. Such candidates might be required to bring along the documents mentioned below:
- Class 10 marksheet
- Class 12 marksheet
- UG marksheets (if applicable)
- UG degree (if applicable)
- Migration certificate
- School leaving certificate/transfer certificate
- Work experience certificate (if applicable)
New Question
11 months agoContributor-Level 10
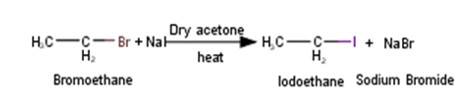
In the presence of sulphuric acid (H2SO4), KI produces HI as follows : -
2KI + H2SO4 → 2KHSO4 + 2KI
Since H2SO4 is an oxidising agent, it oxidises HI (produced in the reaction to I2)
2HI + H2SO4 → I2 + SO2 +;
As a result, the reaction between an alcohol and HI to produce alkyl iodide cannot occur. Therefore, sulphuric acid is not used during the reaction of alcohols with KI. Instead, a non-oxidising acid such as H3PO4 is used in the reaction to get the desired product.
A few things you can remember here, while solving NCERT Solutions for Haloalkanes And Haloarenes.
Sulphuric acid is a powerful oxidising agent. We know this because
New Question
11 months agoContributor-Level 10
Hi, you can refer below for the top Game Designing colleges in India that offer PG programs like MDes, MSc, MA, MFA, ME, MTech, etc.:
| College Name | Tuition Fee | Courses Offered |
|---|---|---|
| Pearl Academy | INR 11.85 lakh | M.Des. in Communication Design |
| WUD | INR 8.05 lakh | M.Des. in Communication Design |
| Amity University Noida | INR 2.88 lakh | M.A. in Game Design & Development |
| SGT University Gurgaon | INR 83,000 | M.Tech. in Computer Science and Engineering (Networking) |
| Jain Deemed-to-be University | INR 6.5 lakh | M.A. in Interactive Game Art Design and Development |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts