Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 10
7.38
The electronegativity of N (3.0) is higher than that of P (2.1). Due to this N-H bond is more polar than a
P-H bond. Also, P and H have the same electronegativity of 2.1 i.e., the P-H bond is non-polar. Therefore, PH3 does not form hydrogen form.
The structure of NH3 and PH3 with their electronegativity is represented below.

New Question
11 months agoContributor-Level 10
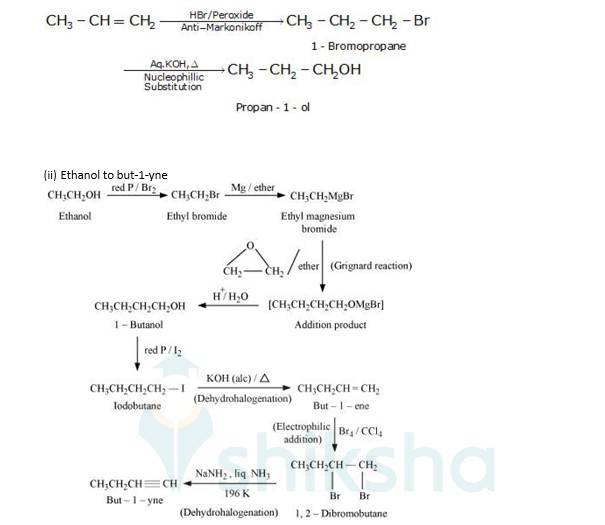
(i) Propene to propan-1-ol
It is an antimarkovnikoff reaction in which under the presence of a peroxide an alkene undergoes substitution, wherein the halo group is attached to that carbon which has least no. Of alkyl groups attached to it.
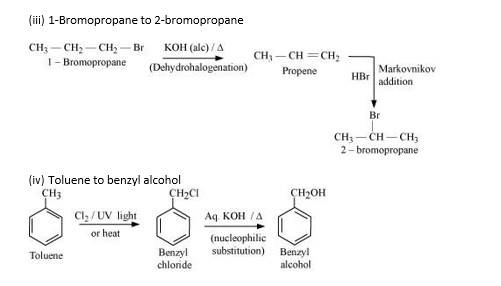
New Question
11 months agoContributor-Level 9
The application process at Siva Sivani Degree College Hyderabad (SSDCH) is conducted in both online and offline modes. Check below the steps for Siva Sivani Degree College Hyderabad (SSDCH) online application process:
Step 1: Visit Siva Sivani Degree College Hyderabad (SSDCH) official website and tap on "Apply Now".
Step 2: Fill the application form with basic details around personal and educational background.
Step 3: Pay the application fee and upload the relevant documents.
Step 4: Submit the form and take a printout.
New Question
11 months agoContributor-Level 10
BDes in Animation is an undergraduate course which continues for 4 year duration in most colleges of India. The admission process for BDes Animation degree requires students to fulfill course eligibility and submit the required documents which are:
- Class 12 Marksheet
- Class 10 Marksheet
- A Valid Id Proof (Aadhar Card, PAN Card, Passport, etc.)
- Passport Size Photo
- Migration Certificate
New Question
11 months agoContributor-Level 10
TalentSprint Hyderabad Certificate fee structure is a composition of multiple fee components. As per the fee structure, students are required to pay three major components during the course duration: tuition fees, a one-time payment and other fees. Combining all, the total fees for the Certificate courses range from INR 1.2 Lacs to INR 9 lakh. Check the below table to learn the component-wise bifurcation of the total fees:
Fee Components | Amount |
|---|---|
Tuition Fees | INR 60,000 - INR 8 lakh |
One-time Payment | INR 10,000 - INR 20,000 |
Other Fees | INR 50,000 - INR 80,000 |
Total Fees | INR 1.2 Lacs - INR 9 lakh |
Note: The above-mentioned fee is as per the official sources. However, it is indicative and subject to change.
New Question
11 months agoContributor-Level 10
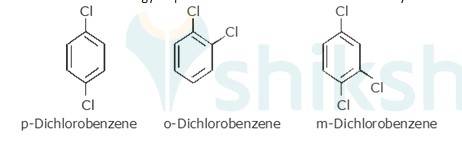
p-Dichlorobenzene is more symmetrical than o-and m-isomers. Because it fits more closely and easily in the crystal lattice than o-and m-isomers. Therefore, more energy is required to break the crystal lattice of p-dichlorobenzene. Therefore, p-dichlorobenzene is symmetrical and has a higher melting point and lower solubility than o-and m-isomers due to strong force of attraction in crystal.
Therefore more energy required to break lattice and will not easily be soluble.
New Question
11 months agoContributor-Level 10
Cluster University MBA admission criteria is entrance-based. The candidates are selected based on performance in the CUET-PG exam. Hence, the aspirants need to apply for CUET-PG and secure a good score. The entrance test applications can be filled out on the official website of National Testing Agency (NTA). Once the CUET-PG results are out, the counselling process commences. During counselling, the seat allotment is done. Selected students can pay the admission fee amount to confirm their seats.
New Question
11 months agoContributor-Level 10
7.37
Chemical reactivity of group 15 elements towards hydrogen, oxygen, halogens, and metals are discussed below.
Reactivity towards hydrogen: Group 15 elements react with H to form hydrides of type EH3 where E=N, P, As, Sb or On moving down from NH3 to BiH3, the stability of the hydrides decreases. For example, the P-H bond in PH3 is less stable than the N-H bond in NH3. The strength of the E-H bond gets weaker as the size of the central atom increases.
Stability order of E-H bond (where E is group 15 elements) can be represented as
N-H > P-H > As-H >Sb-H > Bi-H
Reactivity towards oxygen: Group 15 elements react with O to form oxides
New Question
11 months agoContributor-Level 10
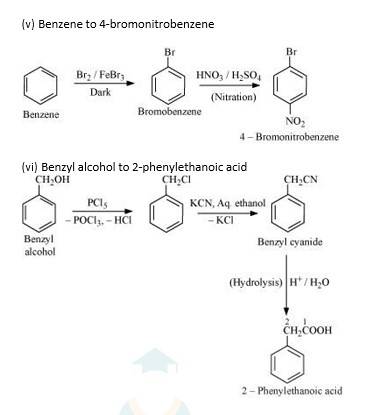
Hydrolysis by KOH results in the formation of the carbocation. Those compounds which leads to the formation of stable carbocation are easily hydrolysed.C6H5CH2Cl leads to formation of 1°- carbocation, while C6H5CHClC6H5 forms 2°-carbocation, which is more stable than 1°-carbocation. Hence C6H5CHClC6H5, is hydrolyzed more easily than C6H5CH2Cl by aqueous KOH.
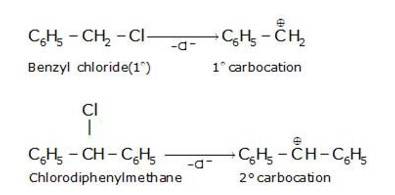
New Question
11 months agoContributor-Level 9
To be eligible for the BCom programme offered at Siva Sivani Degree College Hyderabad (SSDCH), students must have passed Class 10 and Class 12 from a recognised board. As admissions are entirely merit-based, students need not appear for any entrance exams. Eligible candidates can apply for the course through the college's website.
New Question
11 months agoContributor-Level 10
The reaction involves the approaching of the nucleophile to the carbon atom to which the leaving group is attached. When the nucleophile is sterically hindered (does not have any free space or very crowded), then the reactivity towards SN2 displacement decreases. Due to the presence of substituents, hindrance to the approaching nucleophile increases in the following order.
- Bromopentane < 2-bromopentane < 2-Bromo-2-methylbutane. The structures are shown below:
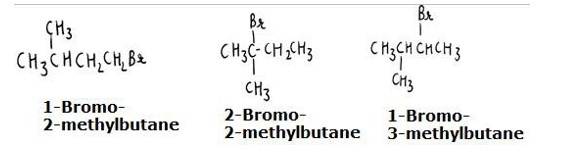
Hence, the increasing order of reactivity towards SN2 displacement is:
- Bromo-2-methylbutane < 2-Bromopentane < 1-Bromopentane
- The stearic hinderance in alkyl halides increases in the order of 1° < 2 < 3, the increasing order of reactivity towards SN2 displacement is given as-
3° < 2 < 1.
The structures are given below:

Hence, the given set of compounds can be arranged in the in
New Question
11 months agoNew Question
11 months agoContributor-Level 10
CII Institute of Hospitality - ITC Kohenur offers admissions mainly on the basis of an aptitude test followed by Industry Panel Viva based on which the seat is offered. Hence, no, the institute does not accept students on a direct basis. However, students interested in joining the institute mau connect with the college via mail or phone to know more about the admission process and chances of getting into the course of choice.
New Question
11 months agoContributor-Level 10
Hi, the top courses offered by best Game Designing colleges are given below:
| Courses Offered | No. of Colleges |
|---|---|
| Certificate | 240+ |
| UG Diploma | 50+ |
| BDes | 30+ |
| BSc | 20+ |
| BA | 10+ |
| After 10th Diploma | 10+ |
| MDes | 9 |
| PG Diploma | 8 |
| MSc | 7 |
| B.E. / BTech | 4 |
| MA | 4 |
Hope this helps!
New Question
11 months agoContributor-Level 10
7.2
Nitrogen atom can bond with another nitrogen atom by strong p–p overlap resulting in NN. The triple bond in N2 has high bond strength resulting in high bond dissociation energy. Phosphorous do not show this property of p–p overlap. Hence, nitrogen is less reactive than phosphorous.

New Question
11 months agoContributor-Level 10
The admission process for the BSc course at AISECT University Hazaribagh and DVRU Bihar is almost similar, in which candidates are required to complete their Class 12 from the recognised board of education in Science stream. Moreover, the selection process is based on merit in both universities. However, DVRU Bihar also accepts CUET UG scores for admissions to BSc courses.
New Question
11 months agoContributor-Level 10
The given reaction is a nucleophillic reaction:

The given reaction is an SN2 reaction. In this reaction, CN acts as the stronger nucleophile and attacks the carbon atom to which Br is attached in nBuBr.
And, CN- ion is an ambident nucleophile and can attack through both C and N. In this case, it attacks through the C-atom.
The mechanism is shown below:
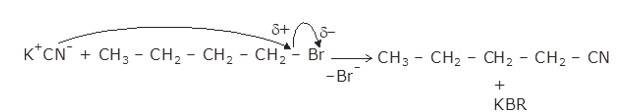
New Question
11 months agoContributor-Level 6
The popular courses at CIIIH ITC Mumbai include the global-standard traditional Swiss Hospitality programs provided in collaboration with Ecole Hôtelière de Lausanne (EHL) School of Hospitality. EHL is the international standard-setter in the field of luxury Hospitality and Hotel Management. Some other courses include the Swiss International Culinary Professional Diploma, the Swiss International Food and Beverage Service Professional Diploma, and the Swiss International Rooms Professional Diploma.
New Question
11 months agoContributor-Level 9
Siva Sivani Degree College Hyderabad (SSDCH) provides the BCom programme in two specialisations, i.e., BCom in Business Analytics, BCom in Computer Applications and BCom (Hons.). Admission to all the specilisations are subject to student's performance in the last qualifying exam i.e., Class 12 or equivalent.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts

