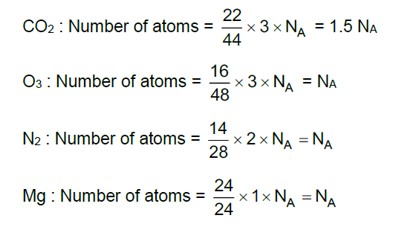Ask & Answer: India's Largest Education Community
All Questions
New Question
6 months agoNew Question
6 months agoBeginner-Level 2
Yes, doing B.E. CSE (Hons.) in IBM Information Security at Chandigarh University is worth it if you're serious about cybersecurity. The course is designed with IBM, so you don't just study theory but also get practical exposure in labs, projects, and case studies. You'll learn things like ethical hacking, cryptography, and how to protect systems from cyberattacks. Since security is a growing concern in every industry, the demand for skilled professionals is high, and having IBM attached to your degree makes your profile stronger. Placements are good, with companies hiring for roles like security analyst, network security engineer, or e
New Question
6 months agoNew Question
6 months agoContributor-Level 10
Haemoglobin is a positive colloid. Hence greater is the charge of anion, more effective will be the coagulation of haemoglobin.
Correct order of coagulating power is
New Question
6 months agoNew Question
6 months agoContributor-Level 10
Electrophoresis : The migration of colloidal particles under the influence of an electric field is known as electrophoresis.
New Question
6 months agoNew Question
6 months agoNew Question
6 months agoBeginner-Level 3
At the same time, general CSE at Chandigarh University is also an equally powerful choice. CU's engineering department is among the largest in India, offering more than 20 CSE specializations, but core CSE still remains the most in demand. The placement record here is outstanding—companies like Microsoft, Amazon, Google, SAP Labs, and Wipro regularly recruit in big numbers. The advantage of general CSE is flexibility.
You'll build a strong foundation in programming, data structures, algorithms, and software development, and later you can branch into AI, cybersecurity, or even Big Data if you want. CU supports this through electi
New Question
6 months agoNew Question
6 months agoContributor-Level 10
Kindly go through the answers
Element | V | Ti | Mn | Sc |
| 515 | 473 | 281 | 326 |
New Question
6 months agoNew Question
6 months agoNew Question
6 months agoNew Question
6 months agoContributor-Level 10
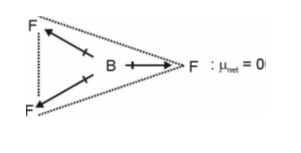
XeF2 : Hybrid orbital = sigma bond + L.P
= 2 + 3
= 5 = sp3d
Shape will be linear
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts