Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type as classified in NCERT Exemplar
Correct Option: (i)
Ans: (i) Conductivity is due to the movement of ions. The number of ions present per unit volume decides the conductivity. It decreases on dilution. The number of ions per unit volume decreases on dilution. So, the assertion is correct, the reason is correct and the reason is the correct explanation of the assertion. Therefore, this option is correct
(ii) Assertion is correct, the reason is correct and the reason is the correct explanation of assertion. Therefore, this option is incorrect
(iii) Assertion and true both are correct therefore this
New Question
10 months agoContributor-Level 7
The admission process for International Institute of Technology and Management comprises different rounds. Students are admitted based on their candidature and performance in the selection rounds. The in-detailed steps for International Institute of Technology and Management admission process are as follows:
- Application Process: The application process is conducted in both online and offline modes. Candidates can visit the college's website or the campus to apply for their desired programme.
- Selection Rounds: International Institute of Technology and Management admissions are both merit-based and entrance-based. For courses such as MBA,
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type as classified in NCERT Exemplar
Correct option: B
Both assertion and reason are valid, but reason does not explain assertion because the order of reaction can be zero or even fractional because the order of reaction is proportional to the total of the power of the reactants.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type as classified in NCERT Exemplar
Ans: Correct Option: (iii)
(i)A chemical reaction is spontaneous when the Gibbs free energy is negative. This is possible when Ecell is positive. Ecathode < Eannode .Therefore, the assertion is correct and the reason is incorrect and the reason is not a correct explanation of assertion. Therefore, this option is incorrect
(ii) Ecathode < Eannode reaction is spontaneous when the Gibbs free energy is negative. This is possible when Ecell is positive. Ecathode < Eannode . Therefore, the assertion is correct and the reason is incorrect. Therefore, this option is co
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type as classified in NCERT Exemplar
Ans: Correct Option: (iii)
(i)The electrode potential of Cu2+/Cu is negative. The electrode potential of 2H+/H2 is 0.00V. Therefore, the assertion is correct and the reason is incorrect and the reason is not a correct explanation of assertion. Therefore, this option is incorrect
(ii) E Cu2+/Cu is not negative so the reason is incorrect. Therefore, this option is incorrect
(iii) The electrode potential of Cu2+/Cu is negative. The electrode potential of 2H+/H2 is 0.00V. Therefore, the assertion is correct and the reason is incorrect. Therefore, this opt
New Question
10 months agoContributor-Level 8
No, you cannot fill the K-LAT application form offline. The process is entirely an online procedure. Applicants must:
- Register and submit the form on the official KIITEE portal (e.g., kiitee.kiit.ac.in or law.kiit.ac.in).
- Upload scanned documents (photograph, signature, academic records) as part of the online submission.?
- Pay any applicable fees online (though typically, no application fee is charged).?
Earlier editions once allowed offline forms, but from recent years, including KIITEE Law 2024/2025, only online applications are accepted.
New Question
10 months agoContributor-Level 10
Yes, the MP Board may revise the syllabus slightly every year. For 2026 exams, students should check the official notification or our website to check if there are any changes. Sometimes, topics are added or removed based on the latest academic updates. If there is any reduction in the syllabus, it will be clearly mentioned by the board. Students should always follow the updated syllabus to prepare effectively. Teachers also help guide students about any changes.
New Question
10 months agoContributor-Level 10
No, there is no negative marking for objective-type questions in the final Goa SSC exams. Each objective question carries one mark, and students are given marks only for correct answers. Incorrect answers do not lead to any deduction of marks, making it advantageous for all the candidates to attempt all questions in the exam. This allows students to take guesses without the fear of losing marks.
New Question
10 months agoContributor-Level 10
This is a Matching Type Question as classified in NCERT Exemplar
(i)- (b) ; (ii)- (a) ; (iii)- (d); (iv)- (c)
The rate rule was discovered experimentally and can be used to predict the reaction rate and reactant concentrations.
The rate constant is the proportionality constant that describes the relationship between the molar concentration of the reactants and the rate of a chemical reaction.
The rate constant (k) of a reaction is proportional to its temperature, i.e., for a given reaction at a given temperature, the rate constant (k) is constant.
The order of a reaction is a quantity that has been empirically determined. As a result
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i) F2 is a non-metal which is the best oxidizing agent since the standard reduction potential of F2 is 2.87V.
(ii)Liis a metal and the strongest reducing agent because the standard reduction potential of Li is −3.05V.
(iii)Au3+ is a metal ion which is an oxidizing agent as standard reduction potential of Au3+ is 1.40V.
(iv) Br−is an anion that can be oxidized by Au3+ as standard reduction potential of Au3+ is 1.40V which is more than that of Br− which is E Br2/Br−0=1.09V.
(v)Auis an unreactive metal
(vi)Li+ is a metal ion having
New Question
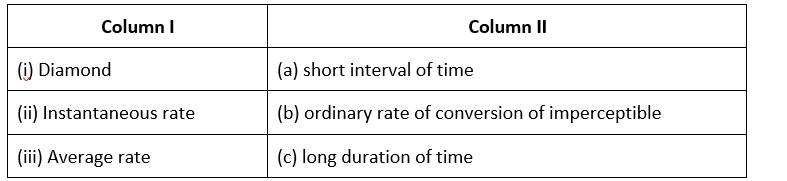
10 months agoContributor-Level 10
This is a Matching Type Question as classified in NCERT Exemplar
(i)- (b) ; (ii)- (c) ; (iii)- (a)
The rate of conversion in diamond is normally imperceptible.
Long-term rate at a moment's notice
The average rate is only for a short time.
New Question
10 months agoContributor-Level 6
The SSC GD score card 2025 was released on June 26, 2025. The Staff Selection Commission (SSC) released the SSC GD scorecard or marks on the official website- ssc.gov.in. Candidates who took the SSC GD 2025 exam can download their score cards by logging to their registered accounts. The direct SSC GD marks 2025 download link will be shared here - https://www.shiksha.com/sarkari-exams/ssc/articles/ssc-gd-marks-score-card-blogId-122595
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)The reaction occurring at Lechlanche cells are
At anode: Zn→Zn2+ + 2e−
At cathode: MnO2 + NH4+ + e−→MnO (OH)+NH3
(ii)Ni−Cd is rechargeable. So it has a longer lifetime.
(iii) Energy in the fuel cell is due to the combustion process. It converts combustion energy into electrical energy 2H2 + O2→2H2O
(iv) Mercury cells don’t involve any solution and are used in hearing aids.
Hence, the answer is:
(i)-d; (ii)-c; (iii)-a, e; (iv)-b
New Question
10 months agoContributor-Level 10
This is a Matching Type Question as classified in NCERT Exemplar
(i)- (c) ; (ii)- (a) ; (iii)- (d); (iv)- (f); (v)- (b); (vi)- (b)
A catalyst can influence the rate of a process by lowering the activation energy
When it comes to molecularity, there can't be a fraction or a zero.
The second half life of a first order reaction is the same as the first.
Activation energy refers to the percentage of molecules with an energy equal to or greater than activation energy.
Correct orientation is not always present when it comes to energetically favourable activities.
Because the area under the Maxwell Boltzmann curve is constant, the total probability
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)Conductivity k is defined as k =
(ii)Molar conductivity is given by Λm =
(iii)Degree of dissociation is given by ? =
(iv)Charge is the product of current and time = I
Hence, the answer is:
(i)-d; (ii)-c; (iii)-b; (iv)-a
New Question
10 months agoContributor-Level 10
Students can download the MPBSE 10th exam syllabus from the official website of the board at mpbse.nic.in. The MP Board 10th syllabus is made available online as a PDF for each paper. We also provide direct links to download the MP Board ryllabus 2026 class 10 on this page. All the schools also provide students the official class 10 syllabus 2026 MPBSE from their respective schools. It's a good idea to check the syllabus at the start of the session. Make sure you are following the latest version released by the board. This will help avoid studying removed or outdated topics.
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i) The cell reaction of a lead storage battery is as follows;
Pb + PbO2 + 2H2SO4→2H2SO4 + 2H2O
At cathode:PbO2 (s)+SO42− (aq)+2e−→2PbSO4 (s) + 2H2O (l)
At anode:Pb (s)+SO42− (aq)→PbSO4 (s)+2e−
Therefore Pb is the anode and PbO2 is cathode
(ii)Mercury cell doesn’t contain ions so it gives steady potential.
(iii)Fuel cells have maximum efficiency as they produce energy due to the combustion reaction of fuel.
(iv)Rusting is prevented by galvanization.
Hence, the answer is:
(i)-d ; (ii)-c ; (iii)-a ; (iv)-b
New Question
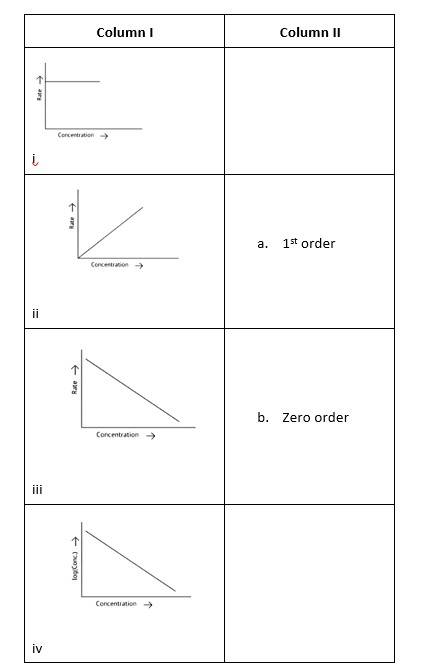
10 months agoContributor-Level 10
This is a Matching Type Question as classified in NCERT Exemplar
(i)- (a) ; (ii)- (b) ; (iii)- (b); (iv)- (a)
A zero-order reaction is one in which the reactant concentrations do not change over time and the rate of concentration remains constant.
A first-order reaction is one in which the rate of the reaction is linearly proportional to the concentration of only one ingredient. In other terms, a first-order reaction is a chemical reaction whose rate is determined by changes in only one of the reactants' concentration.
New Question
10 months agoContributor-Level 10
IAMR Group of Institutions Ghaziabad admissions are generally done on the basis of admission criteria of the desired programme. The institute accepts applicants on both merit and entrance basis depending on the course selected by the students. Hence, although not mandatory, most of the offered courses require students to either appear for institute-level Personal Interview or provide scores obtained in the accepted test. Students may connect with the admission desk of the college to learn more about the courses and their respective admission criteria.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts