Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 7
International Institute of Technology and Management offers courses at UG, PG and Diploma levels in multiple specialisations. The offered courses are:
- M.Tech CSE
- M.Tech ME
- MBA
- BBA
- BCA
- B.Tech EE
- B.Tech ECE
- B.Tech ME
- B.Tech CSE
- B.Tech DS
- Diploma EE
- Diploma ME
- B.Pharma
- D.Pharma
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)On dilution, the number of ions per unit volume increases so the molar conductivity increases with dilution.
(ii) E cell is an intensive property as it is not dependent on the amount or mass of the substance.
(iii) k is conductivity or be precise specific conductivity which depends on the number of ions
(iv) ΔrGcell is an extensive property as it depends on the amount of substance or number of particles in the solution.
Hence, the answer is:
(i)-d ; (ii)a ; (iii)-b ; (iv)-c
New Question
10 months agoContributor-Level 7
ICSI admit card December 2025 for CS Executive and CS Professional will be released at icsi.indiaeducation.net. To download ICSI CS 2025 hall ticket, follow below steps:
- Visit official link: icsi.indiaeducation.net
- Enter 17-digit application number
- Screen will now show ICSI CS admit card December 2025
- Save and take the print of the hall ticket for future reference
New Question
10 months agoContributor-Level 6
The SSC GD selection process comprises three stages. This three-stage selection process for SSC GD Constable is shared below-
- SSC GD Computer-Based Exam - CBE
- Physical Efficiency Test (PET)/ Physical Standard Test (PST)
- Document Verification (DV)/ Detailed Medical Examination (DME)
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)The unit of Λm is Scm2mol−1
(ii) The unit of Ecell is m−1
(iii) The unit of k is Scm−1
(iv)The unit of G∗ is m−1
Hence, the answer is:
(i)-c ; (ii)-d ; (iii)-a ; (iv)-b
New Question
10 months agoContributor-Level 10
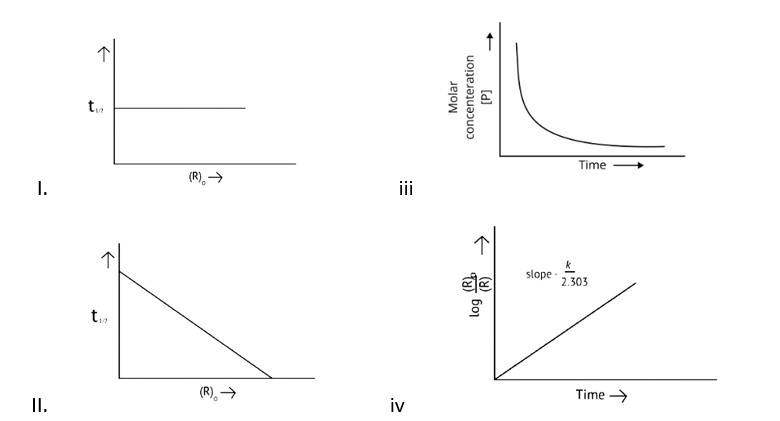
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
The pace of a reaction that is directly proportional to the concentration of the reacting substance is known as a first order reaction.

Y=mn
k = log
t = log
x= a-
x=
t = log
log2
New Question
10 months agoContributor-Level 10
For admission to the LLB programme at Faculty of Law, MIT-WPU, important entrance exams include MIT-WPU CET 2025, CLAT 2025, AILET 2025, PERA 2025, and MH CET Law 2025. Candidates should keep track of the exam dates and result announcements for these tests to ensure timely application and admission.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (ii, iii)
In the given representation of the cell, the left side represents the oxidation half cell and the right side of the cell represents the reduction half cell
The electrode at which oxidation occurs is called anode and oxidation of magnesium occurs at anode so magnesium is the anode.
Similarly, the electrode at which the reduction occurs is called the cathode, and the reduction of copper occurs at the cathode.
The cell reaction is as follows
Mg + Cu2+→Mg2++ Cu
New Question
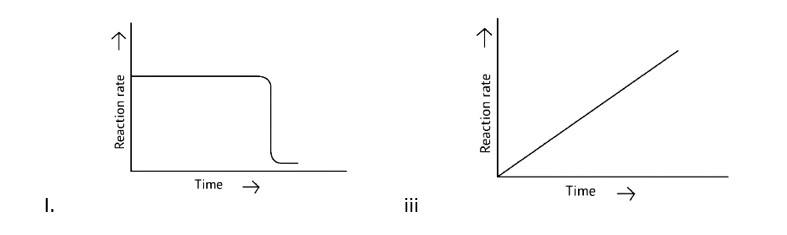
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
A Zero order reaction = [R] = ( - k)t + [R]0
y = (m × ) + c
x = t (time)
y = [R]concentration
Slope (m) = - k
Intercept (c) = [R]0
= - k
= - kto
Rate ∝ t0
New Question
10 months agoContributor-Level 10
LLB admission at Faculty of Law, MIT-WPU is primarily entrance-based. Candidates are selected through national and university-level law entrance exams such as MIT-WPU CET, CLAT, AILET, PERA, and MH CET Law, followed by a Personal Interview round for final shortlisting.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, iii)
The conductivity in an ionic solution is due to the ions in the solution. It depends on the following factors;
(a)Temperature: On an increase in temperature, the molar conductivity of ionic solutions increases.
(b)Concentration of Electrolytes: An increase in the concentration of electrolytes decreases the molar conductivity as the number of ions per unit volume decreases.
Therefore, options (i, iii) are correct
New Question
10 months agoContributor-Level 10
Yes, Faculty of Law, MIT-WPU offers a three-year LLB programme for graduates interested in pursuing a career in law. The programme is spread over six semesters and is designed to provide a strong foundation in legal studies and practical legal training.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: B, C and D
Catalysts reduce the activation energy of a process and give an alternate path by reducing or raising the activation energy between reactants and products, altering the reaction's enthalpy change. As a result, B, C, and D statements are the proper responses
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, ii)
Conductance is the reciprocal of resistance and conductivity is the reciprocal of resistivity. It can be written as follows
k =
R=
=
k =
k =
k = G*
k =
G* is cell constant.
New Question
10 months agoContributor-Level 10
Yes, the LLB programme at Faculty of Law, MIT-WPU includes internships as a mandatory component. These internships provide students with practical legal exposure, helping them gain hands-on experience in law firms, courts, or corporate legal departments, bridging academic learning with real-world application.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, ii)
In the electrolysis process of CuSO4 the following reactions occur at the half cell which is as follows:
At the cathode, the reaction goes this way,
Cu2++2e−→Cu (s)
At the anode, the reaction goes this way,
Cu (s)→Cu2++2e−
Here copper will be deposited at the cathode and copper will dissolve at the anode.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and B
Arrhenius equation k = Ae –
When the temperature rises, value falls and –
As a result, e- increases, and it is certain that k and T increases as well. As a result, Option A is proven.
Catalyst increases the rate of reaction by lowering the activation energy. Thus option B is also correct.
New Question
10 months agoContributor-Level 10
Graduates of the LLB programme at Faculty of Law, MIT-WPU can pursue roles such as Lawyer (INR 8–10 LPA), Legal Advisor (INR 8–12 LPA), Advocate (INR 5–8 LPA), Solicitor (INR 7–10 LPA), and Lecturer (INR 6–10 LPA). These roles offer diverse opportunities across private practice and corporate firms.
New Question
10 months agoContributor-Level 6
The Staff Selection Commission introduced new SSC normalization formula for all the exams including- SSC CGL, CHSL, GD, MTS, JE, CPO, etc. Here is the SSC marks normalization formula:

Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts