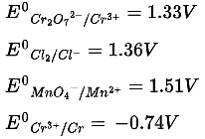Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 9
Yes, University of Rochester is one of the best universities for international students. University ranked #44 among national universities and #27 among best value schools. University is popular for various other reasons discussed below:
- University of Rochester's graduation rate is 77%. This shows the university offers high-quality education and diverse opportunities to students.
- With a 9:1 student-faculty ratio, the university has 68% of classes with fewer than 20 students. This shows the university believes in providing quality education.
- University offers top-ranked programs such as Social Science, Biological and Biomedical Science, He
New Question
10 months agoContributor-Level 9
According to unofficial sources, University of Rochester acceptance rate is 36%. Low acceptance percentage shows that university is hard to get into. Students must perform excellently in academics and be involved in extracurricular activities to make a positive impression. University emphasizes holistic admission review by considering academic records, standardized test scores, essays, recommendations, extracurricular activities and personal qualities
New Question
10 months agoContributor-Level 10
Candidates must check out the selection criteria for the course they wish to apply to, in order to participate in the admission process. The selection criteria for the courses at NICC International College of Design and Technology is that candidates must appear for the NICC Entrance Exam and clear the cutoff that has been set by the institute. Candidates who get the final admission offer can pay the course fee and secure their seat.
New Question
10 months agoContributor-Level 9
Yes, it is worth pursuing BTech at the Accurate Institute of Management & Technology as it is approved by the AICTE and has also been accredited by NAAC. It is a private college and was established in 2006 under the aegis of the Accurate Education & Research Society. Further, AIMT Greater Noida has also been awarded for Excellence in Management Education in North India. The college offers a BTech programme to students across several specialisations.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (ii)
Given: Salt ammonium hydroxide
Apply Kohlrausch Law
Kohlrausch law of states that limiting molar conductivity of any salt species is equal to the sum of the limiting molar conductivity of cations and anions of the electrolyte
Λ0m (NaOH) = Λ0m (NH4+) + Λ0m (OH-) + Λ0m (Na+) + Λ0m (OH-) - Λ0m (Na+)- Λ0m (Cl-)
Λ0m (NH4OH) = Λ0m (NH4OH) + Λ0m (NaOH) – Λ0 (NaCl)
New Question
10 months agoContributor-Level 10
Correct options: A and B
This is a Multiple Choice Questions as classified in NCERT Exemplar
Unimolecular reaction: Unimolecular means relating to a single molecular reaction that describes decomposition of a single reactant.
If product A→ (B)
rate = k [A]
Example Na2O4 (g)→2No2 (g)
Order = 1
Molecularity = 1
The slowest step is the rate determining which determines the Order and Molecularity.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (i)
In the lead storage battery, a reverse reaction occurs and lead sulfate is formed at the anode, and at the cathode, it is converted into lead and lead oxide respectively. The reaction is as follows
At the cathode, the reaction goes this way,
PbSO4 (s)+2e−→Pb (s)+SO42− (aq), Reduction process
At the anode, the reaction goes this way,
PbSO4 (s)+2H2O→PbO2 (s)+SO42−+2H+ +2e, oxidation process
The overall reaction is as follows;
2PbSO4 (s)+2H2O→Pb (s)+PbO2 (s)+4H+ (aq)+2SO42− (aq)
Therefore, at anode lead sulfate is r
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iv)
The cell constant is described as the ratio of the length of the object and the area of cross-section. It can be written as follow
G=
As l and A remains constant for an object it can be inferred that the cell constant for a conductivity cell remains constant for a cell.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
Elementary reaction: It is a reaction that occurs in a single step.
Here we must understand about the Order and Molecularity.
Molecularity of a reaction = number of reacting species (collide to bring a chemical reaction).
Discussing about the Order, Rate law is used in Order and how do we equate shall be seen in the next step
rate = k [A] α [B] β
For a balanced chemical equation of an elementary reaction Order and Molecularity is same and can never be 0.
New Question
10 months agoContributor-Level 10
Remo International College offers merit-based admissions to the BSc course. To get admission into BSc courses, candidates are required to qualify for the minimum eligibility criteria first, which state candidates must complete their Class 12 from a relevant stream with a minimum aggregate of 50%. Further, the college releases the merit of shortlisted candidates according to the scores achieved by the applicants in their higher senior secondary.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct Option: (iii)
Ans: Given: One mole of aluminium
Calculate the number of electrons required to convert Al2O3 to Al
It is the charge required to obtain one mole of aluminium from Al2O3
The dissociation of aluminium oxide is as follows;
Al2O3→2Al3++3O2−
Al3++3e−→Al
Therefore 3Fcharge is required to obtain one mole of aluminum from Al2O3.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (i)
According to the given table, ![]() has the most negative value among the given species. So
has the most negative value among the given species. So
Cr3+ is the most stable oxidized species.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options : A, C and D.
Rate law = Experimental rate of the reaction
Suppose aA + bB→cC + dD
Then rate = k [A] α [B] β
Here instead of using complex reaction we use elementary reaction which gets solved in one step
New Question
10 months agoContributor-Level 9
Accurate Institute of Management & Technology is a private college and was established in 2006 under the aegis of the Accurate Education & Research Society. It is also known as AIMT and is located in Greater Noida. Accurate Institute of Management & Technology courses are offered to students at the UG and PG levels.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iv)
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
According to the electrochemical series, the lower the reduction potential higher is the reducing power.
Therefore, the order of reducing power is Mn2+ < Cl < Cr3+ < Cr
New Question
10 months agoContributor-Level 10
The MPBSE 10th syllabus is the official course outline provided by the Madhya Pradesh Board of Secondary Education for the students who are studying in class 10. It includes all subjects like Hindi, English, Science, Mathematics, and Social Science. The syllabus tells students what topics they need to study during the academic year. It helps in proper planning and preparation for the board exams. Knowing the syllabus also helps students focus only on relevant topics. This improves time management and boosts confidence.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
According to the electrochemical series higher the positive value of standard reduction potential of metal ion, the higher is the oxidizing power
Therefore, MnO4− is the strongest oxidizing agent.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Incorrect order results from a balanced chemical equation. KClO3 + 6FeSO4 + 3H2SO4→KCl + 3H2O + 3Fe2 (SO4)3
This reaction is complex and occurs in several steps. The order is determined by the slowest in the reactions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (ii)
According to the electrochemical series and standard reduction potential of metal, the higher the negative value of standard reduction potential stronger will be the reducing agent
In the given options, the standard reduction potential of chromium has the highest value so it is the strongest reducing agent.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts