Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, iii)
Electrolysis of copper sulfate solution is as follows
CuSO4 ? Cu2+ + SO42−
H2O? H+ + OH−
At the cathode, the reaction goes this way,
Cu2+ + 2e− → Cu; E? Cell = 0.34V
H2O− → H2 E? Cell = 0.00V
At the anode, the reaction goes this way,
2SO42- + 2e−→S2O82− E? Cell =1.96V
2H2O→O2 + 4H+ + 4e− E? Cell =1.23V
The reaction will lower the value of E? Cell is preferred at anode so the second reaction is feasible.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
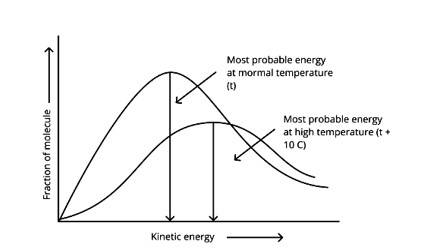
According to the graph, the area under the cure must not vary as the temperature rises.T2 > T1
The energy level of T2 is higher than T1.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, iii)
Kohlrausch law of states that limiting molar conductivity of any salt species is equal to the sum of the limiting molar conductivity of cations and anions of the electrolyte
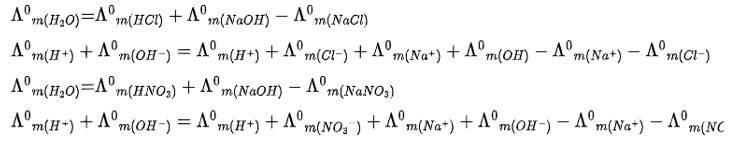
New Question
10 months agoContributor-Level 10
When looking for a college for Medical Transcription course, think practicality. Require colleges that offer real-time transcription practice, and/or have partnerships with hospitals or transcription companies and or services with course upgrades.
Make an observation of student reviews, do a course curricula checks, and look for chunks of experience for.
Also, make sure the institute uses industry standard tools e.g. speech recognition software, and ensure they follow HIPAA regulations, shows me they are training me to be successful.
New Question
10 months agoContributor-Level 10
LLB programme at Faculty of Law, MIT-WPU has a tuition fee of INR 5 Lakh, whereas ILS Law College charges INR 1.1 Lacs and Dr DY Patil Law College charges INR 1.02 Lakh. While MIT-WPU is priced higher, students should also consider return on investment, exposure, and infrastructure.
New Question
10 months agoContributor-Level 10
College ranking matters, but it's not everything for a Medical Transcription program. The important thing is to find a college for Medical Transciption course, that offers practical training, has certified instructors, recognize tools of software, good placement area. Even if a college may not be nationally ranked and considered an elite college, if it has a good record in transcription training and connection with healthcare employers, it's worth observing.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, ii)
Conductivity is due to the movement of ions in the solution. The conductivity of ions depends on the following factors:
(i) nature of electrolyte added
(ii) size of ion produced
(iii) concentration of electrolyte
(iv) nature of the solvent
(v) temperature
Distance between electrodes does not affect the conductivity of an electrolytic solution.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and C
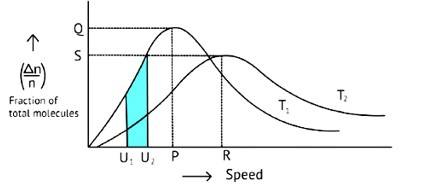
The Maxwell Boltzmann distribution, often known as the Gibbs distribution, is a measure that expresses the likelihood of a system being in each state as a function of the energy of that state.
Look at the graph.
T2 > T1
The fraction of molecules falls as the temperature rises because the area under the curve shrinks.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (ii, iii)
At equilibrium, ?G = -2.303 RT
-nFE = -2.303 RT
E =
For Daniel cell, n=2
E =
At equilibrium, E = 1.1V
1.1V =
(i) Since this option is incorrect
(ii) As derived 1.1V =
(iii) As derived so this option is correct
(iv) As ,so this option is incorrect
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
Activation Energy is the amount of energy released when reactant molecules collide and form an activated complex. When energy is released, the complex decomposes into a product.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, iii)
In the electrolysis of sulphuric acid, the following reactions occur
2SO42− (aq)→S2O82− (aq) + 2e− E? Cell =1.96V
2H2O (l)→O2 (g)+4H+ (aq) + 4e− E? Cell =1.23V
The reaction will lower the value of E? Cell is preferred at anode so the second reaction is feasible.
H+ + e− → H2 E? Cell = 0.00V
At the cathode, reduction of water occurs. Therefore, in dilute sulphuric acid solution, hydrogen will be reduced at the cathode.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A, C and D
The pressure in this reaction is extremely high, and it becomes independent of the ammonia concentration. The metal surface becomes saturated with gas molecules when the rate of reaction=Rate constant. The rate of a zero-order reaction is independent of the concentration of reactants in the reaction.
New Question
10 months agoContributor-Level 6
Sree Ramakrishna Medical College of Naturopathy and Yogic Sciences admission is based on merit scores. No entrance exam is required to secure a seat at the college. The minimum eligibility requires the candidates to pass Class 12 from a recognised board. The admission process also involves counselling and an interview round. Interested candidates can visit the official website to fill out the application form.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (ii, iv)
The lesser the E? value of the redox couple higher is the reducing power
For Cu2+ + 2e−→Cu; E? =0.34V
For 2H+ + 2e−→H2 E? =0.00V
Since the second redox couple has less standard reduction potential than the first so it can be concluded that the redox couple is a stronger oxidizing agent than H+/H2 and copper cannot displace H2 from acid.
New Question
10 months agoContributor-Level 9
An unofficial source claims the University of Rochester acceptance rate is 36%. The low percentage shows that the university is quite stringent towards students during admission and follows a tough admission criterion. The university considers eligibility criteria and academic performance the most crucial factors while choosing deserving applicants. Students must perform exceptionally well in academics and take part in relevant activities, and gain work experience to improve their selection probability.
New Question
10 months agoContributor-Level 10
Before choosing the LLB programme at Faculty of Law, MIT-WPU, students should evaluate factors like curriculum relevance, specialisations offered, faculty experience, internship opportunities, industry exposure, and placement support. Comparing tuition fees, academic reputation, and student reviews also helps make a well-informed decision.
New Question
10 months agoContributor-Level 9
The University of Rochester offers various UG and PG majors for international students. The university is more popular for Social Science, Biological and Biomedical Science, Health Professions, and other professional programs. Some popular University of Rochester majors, along with the graduation rates, are given below:
University of Rochester majors | US News Rankings 2025 |
|---|---|
Social Science | 12% |
Biological and Biomedical Science | 11% |
Health Professions | 11% |
Engineering | 10% |
Business, Management, Marketing | 10% |
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
A complex reaction is one that does not happen in a single step.
The total number of molecules involved in the slowest step of the reaction determines the overall reaction rate.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iv)
The electrolysis of aqueous sodium chloride is as follows
NaCl→Na+ + Cl−
H2O→H+ + OH−
At the cathode, the reaction goes this way,
H2O + e− → H2 + OH−
At the anode, the reaction goes this way,
Cl− (aq)→ Cl2 (g)+ e− E? Cell =1.36V
2H2O (g)→O2 (g) + 4H+ (aq) + 4e− E? Cell =1.23V
At the anode, the reaction with a lower E? value will be preferred and oxidation of oxygen is a slow process and requires high voltage so the first reaction will take place.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts