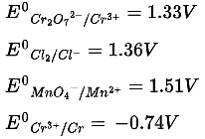Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Simple reactions occur in a single stage, whereas complicated reactions occur in multiple steps. Each phase in a multi-step process has its own molecularity, which is not added together. There is no molecularity in complex reactions. The sequence of a complex reaction is decided by the slowest step. As a result, it can be used for both simple and complex processes
New Question
10 months agoNew Question
10 months agoBeginner-Level 5
With 90 percentile, SEBC girl from Pune, you can get these colleges in CAP Round 1 or 2:
MITAOE Alandi – CS, IT, ENTC
VIT Pune – ENTC, Mech, Civil
DY Patil Akurdi – CS, IT
MMCOE Pune – CS, IT
SKNCOE Vadgaon – CS, IT
PCCOE Nigdi – IT, ENTC (CS in Round 2)
Sinhgad COE – Most branches
COEP & PICT – Very unlikely for CS/IT, try later rounds.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
(i) Conductivity of solution depends upon the size of ions as more is the size of ion, mobility decreases, and conductivity decreases so this option is incorrect.
(ii) Viscosity is the measure of the resistance of a liquid to flow. Greater will be the viscosity of solvent, less will be the flow of electrons and lesser will be the conductivity. Therefore, this option is incorrect.
(iii) Conductivity depends on the solvation of ions in the solution. Greater is the solvation, lesser will be the conductivity of the solution. Therefore, this option
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The number of molecules of the reactants involved in an elementary reaction is referred to as the molecularity of the reaction. For this, a single molecule with a minimum molecularity of one is required, and it cannot be zero
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
On the application of external potential, an increased reaction starts till the opposing voltage becomes 1.1V.
However, further increase in the external potential leads the reaction to start in an opposite direction functioning as an electrolytic cell which is a device that uses electrical energy for carrying out electrochemical reactions.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iv)
(i) Inert electrodes like graphite are a source of electrons in a reaction. It doesn't participate in a chemical reaction. Therefore, this option is incorrect
(ii) Inert electrode chemically doesn't participate in the reaction but it provides a surface either for oxidation and reduction reaction. Therefore, this option is incorrect
(iii) Inert electrodes provide surface for conduction of electrons so this option is incorrect.
(iv) Inert electrodes cease transfer of electrons for oxidation or reduction but not for redox reaction. Therefore
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
The difference between electrode potential potentials of two electrodes when no current is drawn through the cell is termed as cell emf. It is the energy provided by a cell per coulomb of charge passing through it. It is the maximum potential difference between the electrodes of a cell.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
At room temperature, the redox titration of KMnO4vs oxalic acid is exceedingly slow; nevertheless, as the temperature rises, so does the rate of reaction. At a temperature of 500c., KMnO4 is acidified with dil H2SO4 and interacts with oxalic acid.
New Question
10 months agoContributor-Level 10
Ishan Institute of Law is a private institute. The institute is affiliated with CCS University and accredited NAAC A+ Grade. The college is mainly concerned with the Law stream and offers courses such as BA LLB, and LLM. Apart from academics, the college also conducts seminars with fresh contestants, judicial officers, legal officers, academicians, etc.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
(i) The properties which don’t depend on the mass of species are termed intensive properties. ECell is an intensive property that doesn’t depend on the mass of the species.? rG is an extensive property as it depends on the mass of the species. Therefore, this option is incorrect
(ii) ECell is an intensive property but? rG is an extensive property so this option is incorrect
(iii) The properties which don’t depend on the mass of species are termed intensive properties ECell is an intensive property that doesn’t depend on
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Thermodynamic feasibility is determined by ΔG factors. So, while the conversion of diamond to graphite is thermodynamically viable, the reaction is sluggish and has a high activation energy.
Gibb's free energy is ΔG
ΔG =Rtlnk
ΔGP, T o
at constant pressure and temperature should be negative or less than 0.
For example, Diamond to be converted to Graphite
ΔG=−ve
It is theoretically conceivable, but the rate is extremely slow, and the activation energy is extremely high. The energy required to transform diamond to grap
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
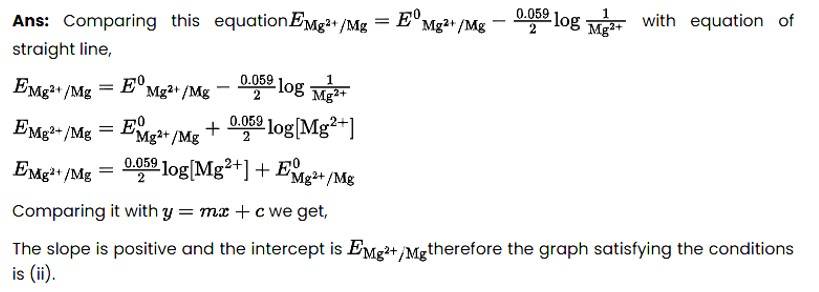
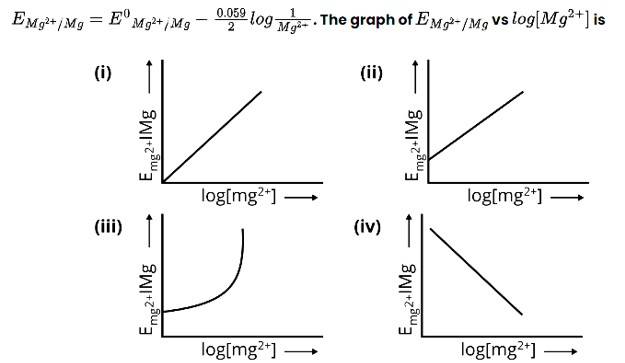
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The pace of any reaction is determined by the concentration of the reactants, the rate of any reaction generally declines during the reaction. As the reaction progresses, the reactant concentration declines, and the product concentration rises.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
(i) For measurement of electrode potential standard conditions must be met which is 1 bar pressure and 1M concentration which is not here so this option is incorrect.
(ii) For measurement of electrodes potential standard conditions must be met which is 1 bar pressure and 1M concentration which is not here so this option is incorrect.
(iii) When copper electrode is connected to SHE it acts as cathode and its standard electrode potential can be measured as follows
E E R- E L
For calculationn of standard potential of a given cell it shoul
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Acetic acid is a weak electrolyte and the number of ions on dilution increases due to an increase in the degree of dissociation. In the case of strong electrolytes the number of ions remains the same but interionic attraction decreases.
New Question
10 months agoContributor-Level 10
Xenon form compounds due to the high electronegativity of fluorine and oxygen, low ionization energy compared to lighter noble gases, and the availability of empty d-orbitals. This breaks the old assumption about noble gases being entirely inert.
New Question
10 months agoContributor-Level 10
To secure a seat in the LLB programme at Faculty of Law, MIT-WPU, candidates must apply online, appear for an accepted entrance exam (MIT-WPU CET, CLAT, AILET, PERA, or MH CET Law), and clear the Personal Interview round. Selection is based on overall performance and seat availability.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts