Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The cell reaction of a lead storage battery is as follows;
Pb + PbO2 + 2H2SO4→2H2SO4 + 2H2O
The density of electrolytes changes because water is formed and sulphuric acid is consumed as a product during discharge of the battery
New Question
10 months agoContributor-Level 10
The LLB programme at Faculty of Law, MIT-WPU has a total of 60 seats available. As admission is limited, it's essential for interested candidates to meet the eligibility criteria and perform well in the required entrance exams.
New Question
10 months agoContributor-Level 7
Yes, admissions are open at International Institute of Technology and Management. Students can apply for their desired UG, PG or Diploma level programmes till Aug 31, 2025 in online/ offline modes.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The chance of more than three molecules meeting at the same time is extremely minimal, reactions with molecularity more than three are extremely rare. Simultaneous collisions decrease as the number grows, the probability is very low, and more than three reactions with molecularity are uncommon.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Primary batteries contain a limited amount of reactants and once consumed are discharged. Secondary batteries take a long time to recharge. Fuel cells run continuously as long as reactants are supplied to them and products are consumed
New Question
10 months agoContributor-Level 10
With the combination of two different halogens, the interhalogen compounds are formed. When compared with pure diatomic halogen molecules, they are more reactive because the bond between different halogens are more polar and weaker. During reactions, it makes them more susceptible to bond cleavage.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The reactions taking place in the Daniel cell is as follows;
Zn+ + Cu2+ → Zn2+ + Cu
The number of electrons involved in two. The Nernst equation is as follows;
Ecell = E cell - log
From the above equation, we conclude that increasing the concentration of Zn2+, Ecell will decrease.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The activation energy for fuel combustion reactions is very large, and because every substance or element requires an ignition temperature to function and excite to a higher temperature, oxygen in air does not combust to fire.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The given cell reaction consists of two half cell reactions represented below as follows;
At anode: Cu→Cu2++ 2e−
At cathode: Cl2 + 2e− →2Cl−
Here oxidation of copper and reduction of chlorine is taking place
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The energy possessed by the reacting species increases as the temperature rises, the pace of a reaction increases. This raises the temperature and raises the average kinetic energy of the reactant molecules
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Standard hydrogen electrode (SHE) is used as a reference electrode and its electrode potential is assumed to be zero. The electrode potential of other electrodes is measured concerning standard hydrogen electrodes.
New Question
10 months agoContributor-Level 10
Due to their high electronegativity, small atomic size, greater ability to form multiple bonds, and absence of d-orbitals, the first elements of each group behave differently.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The reaction's activation energy is relatively high at room temperature and not readily available, the reaction is highly viable. H2 (g) and O2 (g)bond break and clash, allowing particles to pass across the energy barrier.
2H2 + O2→2H2O
Because of the large activation energy, the water molecule does not form in this equation.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Conductivity due to the total number of ions present in the solution is termed as specific conductivity. An increase in the number of ions per unit volume increases. In addition to water, the number of ions per unit volume decreases therefore conductivity decreases.
New Question
10 months agoContributor-Level 10
Jamia Millia Islamia cutoff 2025 has been released across multiple specialisations. Based on Round 1 seat allotment results, BTech cutoff rank stood between 43789 and 256264 for General AII India category students. So, considering Round 1 cutoff, students seeking admission to UNIRAJ must secure over and above cutoff rank 43789 under unreserved quota.
New Question
10 months agoContributor-Level 10
Yes, any degree obtained from IAMR Group of Institutions is valid and widely accepted across the country. The institution is recognised by AICTE, Bar Council of India, IAP, IIC Delhi, NCTE and UGC in addition to being affiliated to various universities, such as AKTU, CCSU, ABVMU, etc. Students can get all the latest information about the college's courses and their degrees by visiting the official website of the institution.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: pH of the substance depends on the concentration of hydronium ions present. Here the concentration of hydronium ions remains constant so pH remains constant.
The electrolysis reaction is as follows;
At anode: 2H2O (l)→O2 (g)+ 4H+ + 4e−
At cathode:4H+ + 4e− →2H2
New Question
10 months agoContributor-Level 10
UK is blessed with a world class educational infrastructure, & is home to some of the top universities in the world. The table below highlights the top 10 universities in the UK per the QS Rankings 2026.
| University | QS University Rank 2026 |
|---|---|
| Imperial College London | #2 |
| University of Oxford | #4 |
| University of Cambridge | #6 |
| UCL | #9 |
| King's College London | #31 |
| The University of Edinburgh | #34 |
| The University of Manchester | #35 |
| University of Bristol | #51 |
| The London School of Economics & Political Science (LSE) | #56 |
| The University of Warwick | #74 |
New Question
10 months agoContributor-Level 10
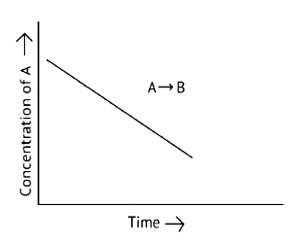
This is a Short Answer Type Question as classified in NCERT Exemplar
A. The reaction has a zero order. The concentration of the reactants does not change with time in a zero order reaction, and the rate of concentration remains constant
B. Slope of the curve is - k
C. The rate constant's units are molL 1s - 1
New Question
10 months agoContributor-Level 10
It is estimated that studying arts colleges in Karnataka is affordable. See the table below for fee ranges of popular Arts colleges in Karnataka:
Fee Range | Top Colleges |
|---|---|
< INR 1 lakh | 2 (St. Joseph's University and St. Aloysius College Magaluru) |
INR 1-2 lakh | 3 (M.S. Ramaiah College of Arts, Science and Commerce, Surana College and St. Claret College) |
INR 3-5 lakh | 3 (Christ University, Mount Carmel College and School of Humanities and Social Sciences, Jain (Deemed-to-be) University) |
INR > 5 lakh | 1 (National School of Journalism and Public Discourse (NSoJ) |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts