Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
William Carey University offers a wide array of clubs, societies, and events designed to enhance student life and foster a vibrant campus community. The university organises various socities for varrious events such as International Afro-Cultural Society, American Chemical Society, Carey Chess Club, Fan clubs, Fellowship of Christian Athletes, and others.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: The relation between Gibbs free energy and the emf of the cell is as follows;
ΔG=−nFEcell
E cell s the cell potential
is the standard emf of the cell
Maximum work obtained from the galvanic cell is nFE .
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Given: Order of the reaction = 0
We know that a zero order reaction is a chemical reaction that occurs regardless of the reactant's concentration.
The rate law of 2A + B→C is
r = k [A]0 [B]0
For a zero- order reaction
r = k
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
A bimolecular reaction occurs when two particles collide. The product of the concentrations of both elements determines the rate of reaction. If one of the reactants is taken in substantial excess in such a way that its concentration seldom changes, a bimolecular reaction can be kinetically first order.
New Question
10 months agoContributor-Level 10
Yes, IAMR Group of Institutions admissions are open for various courses. Interested students can visit the official website of the institute to start the admission process of the desired course. The institutions offers a variety of courses and admits students on the basis of preset admission criteria. In case of any queries, students can connect with the admission desk of the institute via the given contact details.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
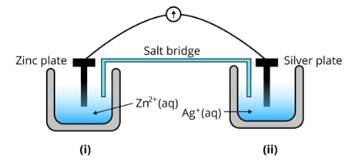
Ans: (i) The diagram is as follows;
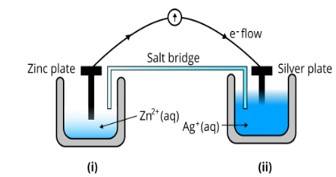
(ii) Agis cathode where the reduction process is taking place where Ag+ takes electrons and deposits them at the cathode
(iii) Potential is zero when the salt bridge is suddenly removed.
(iv) Cell will stop functioning at discharging position when the cell potential is zero
(v) The concentration of Zn2+ ions will increase and the concentration of Ag+ ions will decrease due to conversion in oxidized and reduced forms.
(vi) When the cell is dead, the potential is zero and at equilibrium condition. Thus, the concentration of Zn2+ and A
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Tell us about the pseudo-first-order reaction. It's the reaction with the highest true rate law, yet it behaves like a first order reaction, but it's more specifically a second order reaction.
As an example, consider the hydrolysis of an ester.
CH3COOC2CH5 + H2O→CH3COOH + C2H5OH
rate = k [CH3COOC2H5] [H2O]… (constant)
k1 [CH3COOC2H5]
k = k [H2O]
New Question
10 months agoContributor-Level 10
No, NICC International College of Design and Technology does not offer direct admission. Admission to all the programmess are entrance-based. Candidates are evaluated on their scores in the NICC Entrance Exam. While some colleges in India may offer direct admission through management or NRI quota seats, NICC International College of Design and Technology adheres to a strict entrance based admission process.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Average rate depends upon the change in concentration of reactants or products and the time taken for that change to occur. However, the average rate cannot be used to predict the rate of reaction at a particular instance as it would be constant for the time interval for which it is calculated.
So, to express the rate at a particular moment of time we determine the instantaneous rate. Itis obtained when we consider the average rate at the smallest time interval say dt (i.e. when? t approaches zero).
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
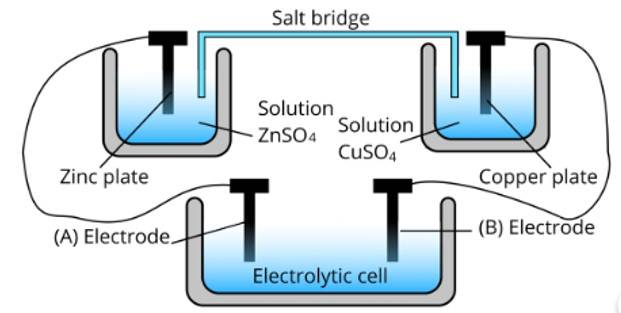
Ans: (i) Cell ‘B’ will act as an electrolytic cell because to less value of Ecell The reactions occurring in the cell are as follows;
At anode: Zn2+ + 2e− → Zn
At cathode: Cu (s) → Cu2+ + 2e−
(ii) Cell ‘B’ has a higher emf so it acts as a galvanic cell. The reactions are as follows;
At anode: Zn → Zn2+ + 2e−
At cathode: Cu2+ + 2e− → Cu
New Question
10 months agoContributor-Level 10
William Carey University offers a range of programs, but the quality and resources can vary. The university is noted for offering quality education at relatively affordable fees. The curriculum is generally considered to be relevant to the needs of the job market and future careers. The university provides a conducive environment for learning and academic growth. While, some students have mentioned concerns about the infrastructure, particularly the limited space and lack of recreational areas.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Ans: Catalyst action is a complicated concept. A catalyst aids in the formation of temporary bonds during a chemical reaction. The objective of a catalyst is to lowers the activation energy. A catalyst is a substance that increases the rate of a reaction without itself undergoing any permanent chemical change. The action of the catalyst can be explained by intermediate complex theory. According to this theory, a catalyst participates in a chemical reaction by forming temporary bonds with the reactants resulting in an intermediate complex. This has transitory existe
New Question
10 months agoNew Question
10 months agoContributor-Level 7
With the release of Round 1 seat allotment results for admission to Mizoram University for All India quotas. Considering the Mizoram University JEE Main cutoff 2025, the closing rank stood at 60031 for B.Tech. in Computer Engineering under General AI quota, which is also the lowest rank to get admission at the institute. It is mandatory for the candidates to acquire an eligible rank in JEE Main to secure a seat.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Ans:
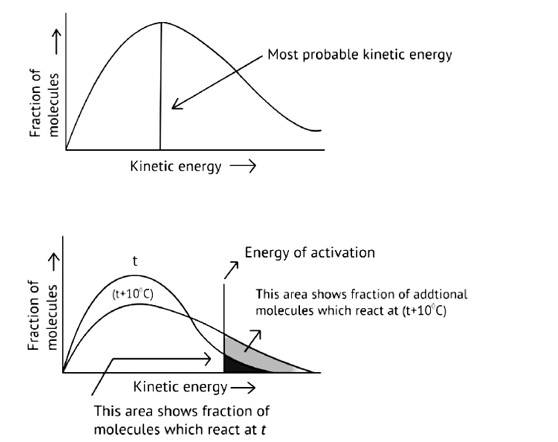
As illustrated in the graph, as the temperature rises, the peak pushes ahead, increasing probable kinetic energy while decreasing the number of molecules utilizing it, resulting in a faster rate of reaction.
New Question
10 months agoContributor-Level 10
According to India Today ranking 2024, Christ University emerged to be as the No. 1 ranked Arts college in Karnataka secured 15th position followed by other colleges like Mount Carmel College and St. Joseph's University.
New Question
10 months agoContributor-Level 10
About 15+ top ranked Arts colleges in Karnataka. Of these, 13 colleges are privately owned. Students can secure admission based on their merit. Some of the best BA colleges in Karnataka include Christ University, Mount Carmel College, St. Joseph's University, Kristu Jayanti College, St. Aloysius College Magaluru, Maharani Lakshmi Ammanni College for Women, The Oxford College of Arts, M.S. Ramaiah College of Arts, Science and Commerce, and many others.
New Question
10 months agoContributor-Level 10
Out of 6 eligible students, 1 MSc Chemistry student was placed during NIT Kurukshetra placements 2025. The salary package offered was 6.5 LPA.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts