Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In the electrolysis process of sodium chloride, oxidation of water at anode requires more potential. So Cl− is oxidized at anode instead of water.
New Question
10 months agoContributor-Level 7
Candidates seeking for admission to International Institute of Technology and Management must meet the course-specific educational requirements defined by the college. Given below is the course-wise eligibility and selection criteria for International Institute of Technology and Management:
| Course Name | Eligibility | Selection Criteria |
|---|---|---|
| BTech | Pass Class 12 from a recognised board | JEE/ CUET |
| MTech | Graduate with BTech from a recognised university | Merit-based |
| MBA | Pass graduation from a recognised university | CAT/ XAT/ MAT |
| BBA | Pass Class 12 from a recognised board | BCA |
| BPharma | Pass Class 12 from a recognised board | Merit-based |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Galvanic cell consists of two electrodes in which anode and cathode are present as electrodes. The anode is present on the left side at which oxidation occurs. The cathode is present on the right side at which reduction occurs and in the middle, there is a salt bridge which is depicted by parallel lines. Therefore, the galvanic cell is Cu|Cu2+ |Ag+ |Ag.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The concentration of the reactants does not change with time in a zero- order reaction, and the rate of concentration remains constant.
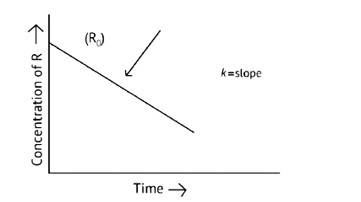
R= [R]0 – Kt
Kt = [R]0
t =
New Question
10 months agoContributor-Level 10
As per India Today ranking 2024, Christ University emerged to be as the top ranked Arts college in Karnataka. Listed below are the colleges along with their rankings 2024:
Disclaimer: This information is sourced from the official website of the ranking body listed and may vary.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: According to Faraday’s second law of electrolysis, the amount of different substances liberated keeping the electricity flow same through the electrolytic solution is directly proportional to the equivalent weight.
=
E1and E2 have different values. Therefore, the mass of copper and silver deposited will be different
New Question
10 months agoContributor-Level 10
The Goa SSC Class 10 syllabus is vital when it comes to passing the class 10 exam of the Goa Board. The syllabus comprises topics and sub-topics for each subject. Questions are asked from the topics included in the syllabus. Knowing the Goa SSC 10th syllabus can be really helpful in scoring well in the exams. Knowing the syllabus in advance also help students prepare a daily study plan. Students will also be able to know which topics require more attention to perform well in the exam.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The concentration of the reactants does not change with time in a zero- order reaction, and the rate of concentration remains constant.
New Question
10 months agoContributor-Level 10
The Anglia Ruskin University wolrd ranking is #501-550 as per QS WUR by Subject 2025. Quacquarelli Symonds (QS) has also ranked the institute for a number of other categories, as mentioned below-
| Category | QS Ranking |
|---|---|
| World University Ranking | #501-550 |
| Sustainability Ranking | #1101-1150 |
| Full-time MBA Rankings-Europe | #70 |
| Full-time MBA Rankings-Global | #201-250 |
New Question
10 months agoContributor-Level 10
The eligibility criteria for the courses at NICC International College of Design and Technology are as follows:
MBA: Candidates must have completed their undergraduation.
BSc: Candidates must have cleared Class 12 with relevant subjects.
UG Diploma: Cadidates must have completed Class 12.
Candidates who meet the above mentioned eligibility criteria are eligible to participate in the admission process.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: At the stage of equilibrium E? Cell =0. So according to the relation,
ΔrG =−nFEcell
ΔrG =−n×F×0
ΔrG =0
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Let order of reaction be = n
rate = k [A]n- (i)
27r = k [3A]n- (ii)
Divide (ii) by (i)
= k [3A]nn
3n = 27
n = 3
New Question
10 months agoContributor-Level 10
For LLB admission at Faculty of Law, MIT-WPU, students need documents like entrance exam scorecard, graduation marksheets, transfer certificate, photo ID, caste certificate (if applicable), passport-sized photos, and proof of address. These must be submitted during the counseling or document verification process for final admission.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: E? Cell can never be zero. For a feasible reaction E? Cell should be positive or ΔrGshould be negative and at the stage of equilibrium, both of these parameters are zero.
New Question
10 months agoContributor-Level 10
The final step of admission to the LLB programme at Faculty of Law, MIT-WPU involves attending a counseling session and paying the tuition fee. The total course fee is INR 5 Lakh, which must be paid according to the university's prescribed schedule and guidelines after confirmation.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The order and molecularity of elementary reactions are the same. As a result of a single collision between two molecules or ions, a complicated process occurs. A reaction mechanism is a set of elementary reactions.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans:No, the difference in potentials of the electrodes is measured. A reference electrode is to be taken while measuring the electrode potential of the electrode.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Experimentally, the reaction's rate law can be calculated.
Step 1
2NO (g) + O2 (g)→2NO2 (g)
O2 (g) is taken in excess
x molecules of NO number reacting with excess of O2
r = k [NO]x
Step 2
2NO (g) + O2 (g)→2NO2 (g)
When 2No (g)is taken in excess
Y molecules of O2 reacting with excess of [NO]
rate = k [O2]y
rate = k [NO]x [NO]y
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
