Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Stronger electrolytes undergo cent percent dissociation. Electrolyte B will be the stronger electrolyte since the concentration of ions remains the same upon dilution.
New Question
10 months agoContributor-Level 7
The application process for International Institute of Technology and Management is conducted in both online and offline modes. Students willing to apply through online mode can check the steps below:
- Visit the official website at International Institute of Technology and Management.
- Tap on the "Application Form" that appears on the Home Page.
- Fill the form with details around your personal and academic background.
- Complete the form and press the "submit" button.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The number of ions present in the cell decides the life-time of any cell. In mercury cells, ions are not involved so the mercury cell has a constant cell potential throughout its useful life.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
LLB programme at Faculty of Law, MIT-WPU includes subjects like Constitutional Law, Indian Penal Code, Criminal and Civil Procedure, Property Law, Consumer Protection, and Legal Research. The curriculum also features legal drafting, internships, forensic science, and interdisciplinary courses like philosophy and communication for a well-rounded legal education.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Electrolysis of brine solution is as follows
NaCl (aq)→Na+ (aq)+Cl− (aq)
At the cathode, the reaction goes this way,
H2O (l) + e−→12H2 (g)+OH− (aq)
At the anode, the reaction goes this way,
Cl− (aq)→12Cl2 (g) + e−
The pH of the solution will increase as sodium hydroxide is being formed.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: When the opposing potential becomes equal to the electrical potential there is no current flowing in the cell and the cell reaction stops and there is no chemical reaction in the cell
New Question
10 months agoContributor-Level 7
International Institute of Technology and Management accepts entrance exams such as JEE Main, CUET, CAT, MAT and CMAT for admission to BTech, BCA, BBA and MBA courses. Students need to qualify these exam with a good score and mentioned the score while filling the application form.
New Question
10 months agoContributor-Level 10
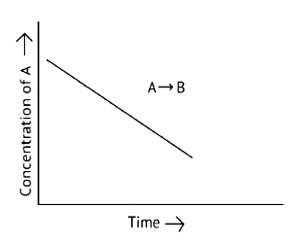
This is a Short Answer Type Question as classified in NCERT Exemplar
The concentration of the reactants does not change with time in a zero -order reaction, and the rate of concentration remains constant. The molecularity of a reaction cannot be 0 because it indicates the number of reactants involved.
New Question
10 months agoContributor-Level 10
Yes, joining the top ranked Arts colleges in Karnataka can be worthwhile for you. For that, you need to check its ROI. Listed below are some colleges along with their tuition fees and average placement package:
College Name | Total Tuition Fee | Average Placement Package |
|---|---|---|
School of Humanities and Social Sciences Courses | INR 4.50 lakh | INR 7.55 LPA |
Christ University Courses | INR 3.21 lakh | INR 4.80 LPA |
| Surana College Courses | INR 1.26 lakh | INR 3 LPA |
Mount Caramel College Courses | INR 3.04 lakh | INR 3.33 LPA |
St. Aloysius College Magaluru Courses | INR 23,100 | INR 3 LPA |
Disclaimer: This information is sourced from the official website and may vary.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Alternating current is used to stop electrolysis so that the concentration of the ions in the solution remains constant.
New Question
10 months agoContributor-Level 10
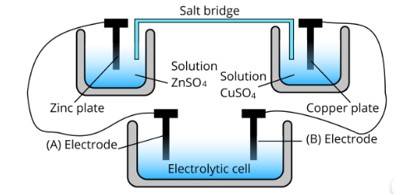
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The cell represented above shows an electrochemical cell in which two different electrodes are present and the cell at the bottom represents the electrolytic cell.
In this zinc is losing electrons moving towards electrode A and copper is accepting an electron from electrode B. Therefore, the polarity of electrode A is positive and electrode B is negative.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
When the Threshold energy is less than the energy of the molecules and the orientation of the molecules is proper for the effective collision, the reaction happens. As a result, the reaction is slow, and the number of effective collisions is reduced
New Question
10 months agoContributor-Level 10
Yes, Remo International College does offer internships to BSc students. Moreover, as per the information sources on the official website of the college, it explicitly shows it offers a live internship programme for students to give them practical exposure, implant training, industrial visits, etc. The college ensures that students get great exposure to aviation sectors and guides them toward job placements.
New Question
10 months agoContributor-Level 10
There are about 10+ best private Arts colleges in Karnataka. See the table below showing private colleges with their total tuition fee and ther India Today ranking 2024:
Private Colleges | Total Tuition Fee | India Today 2024 |
|---|---|---|
Christ University Admission | INR 3.21 lakh | 15 |
St. Joseph's University Admission | INR 90,000 | 18 |
Mount Caramel College Admission | INR 3.04 lakh | 17 |
St. Aloysius College Magaluru Admission | INR 23,100 | 53 |
M.S. Ramaiah College of Arts, Science and Commerce Admission | INR 1.10 lakh | 77 |
Disclaimer: This information is sourced from the official website and may vary.
New Question
10 months agoContributor-Level 10
We cannot say that the Goa SSC syllabus is tough. It is also not considered as the easiest syllabus. the difficulty level will be different for different students.They are advised to prepare all the topics included in the Goa 10th syllabus chapters to secure good marks in the exam. Board exam Questions will be from the Goa SSC syllabus.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The potential difference between the metal and its solution is termed electrode potential.
New Question
10 months agoContributor-Level 10
VUB is partnered with many housing websites in Brussels that provide off-campus accommodations similar to those on campus. These resources are as follows:
- MyKot
- Brik
- Bru Upkot
- U-Residence
- Brusello-XL
- Jet Upkot
- Social rental office SVKS
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The number of ions or atoms that collide to react is known as molecularity in an elementary reaction. The order of reaction about B should be 1 if this is an elementary reaction, however the rate law given is
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts